UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the Month of October 2021
Commission File Number: 001-39621
OPTHEA LIMITED
(Translation of registrant’s name into English)
Level 4
650 Chapel Street
South Yarra, Victoria 3141
Australia
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
EXHIBITS
| Exhibit | Description | |
| 99.1 | Opthea Chairman’s Address to the 2021 Annual General Meeting | |
| 99.2 | ||
| 99.3 | ||
| 99.4 |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| OPTHEA LIMITED |
| Date: October 19, 2021 | By: | /s/ Megan Baldwin | ||||
| Megan Baldwin, Ph.D. | ||||||
| Chief Executive Officer and Managing Director |
Exhibit 99.1

ASX and Media Release
19 October 2021
Opthea Chairman’s Address to the 2021 Annual General Meeting
Melbourne, Australia; 19 October 2021 – Opthea Limited (ASX:OPT; Nasdaq:OPT),
Good morning to those of you who have joined us from Australia and good afternoon to our US shareholders.
Despite the unparalleled circumstance that our nation, our industry, and our company has faced over the past year against the backdrop of a global pandemic, Opthea has advanced significantly. Today, at our Annual General Meeting we are pleased to share our achievements over the past 12 months and update you on our strategic goals and objectives for the current fiscal year.
At Opthea we are forging a new but important path for wet AMD therapies. Whilst many patients administered anti-VEGF-A therapies experience stabilization or improvement in visual acuity, a majority of patients fail to achieve 20/40 or better vision after 12 months of treatment; and many are unable to resume routine daily activities such as driving and reading. We believe in the potential of OPT-302, a novel and unique molecule, which by blocking VEGF-C and VEGF-D, may address limitations in the efficacy of current treatments for wet AMD and other retinal diseases.
So, as we continue on our development path we are motivated by:
| • | Most notably the need to improve outcomes for patients with retinal eye diseases; |
| • | And that there have been no new therapies targeting novel mechanisms approved for wet AMD since the approval of the first VEGF-A inhibitor for wet AMD over 15 years ago. If successfully developed, OPT-302 may change that unfortunate circumstance. |
We are encouraged in our approach because of the sound scientific rationale for targeting VEGF-C and VEGF-D, which is supported by our Phase 2b trial outcomes demonstrating superior vision gains in patients receiving combination OPT-302 therapy.
From an operational perspective, I am pleased to report that Opthea has continued to successfully advance its two large Phase 3 pivotal registrational clinical trials for OPT-302 in wet AMD.
Building on successful End-of-Phase 2 and Scientific Advice regulatory meetings with the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) respectively, we finalized the Phase 3 development pathway for OPT-302 and in March this year, initiated two global, multi-center, randomized, sham-controlled Phase 3 clinical trials, ShORe and COAST. Over an approximate 18-month period, both trials will each enroll approximately 990 patients from more than 20 countries worldwide.
Earlier in the year the FDA granted Fast-Track status to OPT-302 for the treatment of wet AMD – an acknowledgement of the unmet medical need in the clinical management of wet AMD and the potential of OPT-302 to address that unmet medical need.
Our objective is to report Phase 3 topline data in second half of calendar year 2023.
Level 4, 650 Chapel Street, South Yarra, Victoria 3141 Australia T +61 (3) 9826 0399 F +61 (3) 9824 0083
www.opthea.com ABN 32 006 340 567
From a corporate perspective, we completed a U.S. initial public offering (IPO) and NASDAQ listing in October 2020, raising US$128.2 million (AU$164.9 million) equity capital.
Additionally, we have strengthened the board. In addition to my appointment as Chairman of the Board in October 2020 we expanded its strengths in clinical and commercialization strategy with the appointments of Dr. Julia Haller and Ms. Judith Robertson as independent non-executive directors. In addition to the board appointment, we are building out our U.S. team with the intent to position the Company for success and increased shareholder value.
Looking ahead, Opthea will focus over the next 12 months on the execution of the Phase 3 pivotal program for OPT-302 in wet AMD and ensuring the company has the resources and capability to take full advantage of the opportunities we believe this unique program offers.
On behalf of the board and management we would like to thank our shareholders for their support and encouragement. We look to the future with enthusiasm and a single-minded dedication to the objective of delivering high value, both to families of those with disorders of the eye and to our shareholders.
Thank you for your support and investment in Opthea.
About Opthea
Opthea (ASX:OPT; Nasdaq:OPT) is a biopharmaceutical company developing novel therapies to address the unmet need in the treatment of highly prevalent and progressive retinal diseases, including wet age-related macular degeneration (wet AMD) and diabetic macular edema (DME). Opthea’s lead product candidate OPT-302 is in pivotal Phase 3 clinical trials and being developed for use in combination with anti-VEGF-A monotherapies to achieve broader inhibition of the VEGF family, with the goal of improving overall efficacy and demonstrating superior vision gains over that which can be achieved by inhibiting VEGF-A alone.
Inherent risks of Investment in Biotechnology Companies
There are a number of inherent risks associated with the development of pharmaceutical products to a marketable stage. The lengthy clinical trial process is designed to assess the safety and efficacy of a drug prior to commercialization and a significant proportion of drugs fail one or both of these criteria. Other risks include uncertainty of patent protection and proprietary rights, whether patent applications and issued patents will offer adequate protection to enable product development, the obtaining of necessary drug regulatory authority approvals and difficulties caused by the rapid advancements in technology. Companies such as Opthea are dependent on the success of their research and development projects and on the ability to attract funding to support these activities. Investment in research and development projects cannot be assessed on the same fundamentals as trading and manufacturing enterprises. Therefore, investment in companies specializing in drug development must be regarded as highly speculative. Opthea strongly recommends that professional investment advice be sought prior to such investments.
Forward-looking statements
Certain statements in this announcement may contain forward-looking statements, including within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. Any statement describing Company goals, expectations, intentions or beliefs is a forward-looking statement and should be considered an at risk statement, including, but not limited to, the continuation of patient recruitment for Opthea’s pivotal Phase 3 clinical trials of OPT-302 in wet AMD. Such statements are based on Opthea’s current plans, objectives, estimates, expectations and intentions and are subject to certain risks and uncertainties, including risks and uncertainties associated with clinical trials and product development and the impact of general economic, industry or political conditions in Australia, the United States or internationally. These and other risks and uncertainties are described more fully in the section titled “Risk Factors” in the final prospectus filed with the SEC on October 19, 2020. The Company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events, or otherwise, except as required under applicable law. You should not place undue reliance on these forward-looking statements as predictions of future events, which statements apply only as of the date of this announcement. Actual results could differ materially from those discussed in this ASX announcement.
Level 4, 650 Chapel Street, South Yarra, Victoria 3141 Australia T +61 (3) 9826 0399 F +61 (3) 9824 0083
www.opthea.com ABN 32 006 340 567
Authorized for release to ASX by Megan Baldwin, CEO & Managing Director
| Company & Media Enquiries: | Join our email database to receive program updates: | |
| U.S.A. & International: Sam Martin Argot Partners Tel: +1 212-600-1902 opthea@argotpartners.com
Australia: Rudi Michelson Monsoon Communications Tel: +61 (0) 3 9620 3333 |
Tel: +61 (0) 3 9826 0399 info@opthea.com www.opthea.com | |
Level 4, 650 Chapel Street, South Yarra, Victoria 3141 Australia T +61 (3) 9826 0399 F +61 (3) 9824 0083
www.opthea.com ABN 32 006 340 567
Exhibit 99.2

Leading Therapeutic Innovation in Retinal DiseasesOPTHEA.COM | @OptheaLimited | NASDAQ (OPT): ASX (OPT.AX)

DisclaimerThis presentation includes general background information about the activities of Opthea Limited (ABN 32 006 340 567) (‘Opthea’ or ‘Company’) and its affiliates and subsidiaries (together, the ‘Opthea Group’). The information contained in this presentation is in summary form and does not purport to be complete.This presentation contains forward-looking statements within the meaning of the U.S. federal securities laws that involve substantial risks and uncertainties. All statements, other than statements of historical facts, contained in this presentation, including statements regarding our strategy, future operations, future financial position, future revenues, projected costs, prospects, plans and objectives of management, are forward-looking statements. The words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “predict,” “project,” “target,” “potential,” “will,” “would,” “could,” “should,” “continue,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements as predictions of future events. Actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements we make. The forward-looking statements contained in this presentation reflect our current views with respect to future events, and we assume no obligation to update any forward-looking statements except as required by applicable law. Please refer to information, including risk factors, set forth in Opthea’s filings with the U.S. Securities and Exchange Commission for key factors that could cause actual results to differ materially from those projected in the forward-looking statements contained herein. This presentation includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies conducted by third parties as well as our own estimates of potential market opportunities. All of the market data used in this presentation involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such data. Industry publications and third party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. Our estimates of the potential market opportunities for our product candidates include several key assumptions based on our industry knowledge, industry publications, third-party research and other surveys, which may be based on a small sample size and may fail to accurately reflect market opportunities. While we believe that our internal assumptions are reasonable, no independent source has verified such assumptions.The information contained in this presentation does not constitute investment or financial product advice (nor taxation or legal advice) and is not intended to be used as the basis for making an investment decision. It does not take into account the investment objectives, financial situation, taxation position or needs of any particular investor, which should be considered when deciding if an investment is appropriate. You must consider your own investment objectives, financial situation and needs and conduct your own independent investigations and enquiries, including obtaining taxation, legal, financial or other professional advice in relation to the information contained in this presentation as appropriate to your jurisdiction. This presentation should not be relied upon by the Recipient in considering the merits of any particular transaction.This presentation may contain trade marks and trade names of third parties, which are the property of their respective owners. Third party trademarks and trade names used in this presentation belong to the relevant owners and use is not intended to represent sponsorship, approval or association by or with any of the Opthea Group.2

First and only retina asset with strong clinical evidence of better visual outcomes over anti-VEGF-A therapy for wet AMD, with well tolerated safety profileCurrently available treatment options and those in development focus only on OPT-302 has the potential to reducing burden of care, OPT-302 is designed to transform patient revolutionize the treatment outcomes by improving visionof wet AMD and improve OPT-302 expected to be rapidly adopted by patients, physicians and payers and preserve vision for globally due to: millions of patients• High unmet need• Established wet AMD market and clinical practice• Favorable physician and health system economicsWe are developing OPT-302, a FDA granted Fast-Track designation based on superior Phase 2b results first-in-class VEGF-C/D ‘trap’, to be used in combination with standard Pivotal Phase 3 trials ongoing, on-track for topline data 2H 2023 and of care anti-VEGF-A therapies commercial launch 2024OPT-302 represents a multi-billion dollar commercial opportunityLong-term value opportunity substantial:• Composition of Matter and Methods of Use Patents through 2034• Further opportunity for Patent Term Extension (PTE), Data and Market Exclusivity periods beyond 2034• Expansion in to DME, RVO and PCV represent blockbuster upside opportunity3

Opthea’s Experienced Leadership TeamManagement Team Non-Executive Board MembersMegan Karen Adams Jeremy Levin, DPhil, MB BChir Daniel Spiegelman, BA, MBA Baldwin, PhD VP Finance & Chairman Director, Chair Audit & Risk Committee CEO & Managing Company • Appointed Chairman in October 2020 • Appointed in September 2020Director (1) Secretary• Selected Experience: Has extensive experience in • Selected Experience: Former Exec Vice President, the global biopharma industry. Currently CEO, CFO and member of the Board of Directors of Chairman and Founder of Ovid Therapeutics. Biomarin Pharmaceutical Inc. Has provided • Joined Opthea in 2008 and has been Chief • VP Finance & Company Secretary since Formerly President & CEO of Teva Pharmaceutical strategic financial management and insight to life Executive Officer and Managing Director April 2021 Industries Ltd, and Senior Vice President of sciences companies, and previously held roles at since February 2014 • Selected Experience: Directed financial Strategy, Alliances & Transactions at Bristol Myers Genentech, including Treasurer, and CFO of CV • Selected Experience: Over 20 years of operations at several ASX-Nasdaq listed Squibb. Joined BMS from Novartis where he was Therapeutics. Currently serves as a director of experience focusing on angiogenesis and companies. Formerly CFO of Victor Global Head of Strategic Alliances. Has served on several companies, including Myriad Genetics, Jiya therapeutic strategies for cancer and Smorgon group and Director Finance the board of directors of various public and private Acquisitions Corp. and Spruce Bioscience. ophthalmic indications; prior to Opthea, was Nexvet Biopharma and Financial companies, including Biocon Ltd, and is currently employed at Genentech (now Roche) in the Controller of Biota Pharmaceuticals, both on the Board of Lundbeck. US in market planning and research roles. companies having listed on the Nasdaq Holds a PhD in Medicine, having conducted and subsequently been acquired. Julia Haller, MD Judith Robertson, BA, MBA doctoral studies on the biology of VEGF-C and VEGF-D. Director Director• Appointed in June 2021 • Appointed in June 2021• Selected Experience: Internationally recognized • Selected Experience: Commercial executive with ophthalmologist and vitreoretinal surgeon. track record of leading commercial organisations and Ophthalmologist-in-Chief and Endowed Chair at Wills launching multiple ophthalmic products. Previously Eye Hospital in Philadelphia. Professor and Chair of Chief Commercial Officer of Aerie Pharmaceuticals, Mike the Department of Ophthalmology at the Sidney Vice President Immunology and Ophthalmology Clare Price Kimmel Medical College at Thomas Jefferson Global Commercial Strategy Leader at Johnson & Gerometta, PhD Director of Clinical University, and a director Bristol Myers Squibb. Johnson and Opthalmology Global Business Head of CMC Development Previously a director of Celgen Corporation and Franchise Head at Novartis (formerly Alcon). Has also Development Professor of Ophthalmology, Johns Hopkins held roles in sales and marketing at Novartis, Bristol University School of Medicine, The Wilmer Eye Myers Squibb and Searle USA.Institute. Dr Haller received a BA from Princeton, graduating magna cum laude, and completed her • Head of Chemistry, Manufacturing and • Director of Clinical Development since July medical training at Harvard Medical School. Controls Development since December 2016 2008 • Selected Experience: Previously Director • Selected Experience: Has over 30 years of Clinical Development at Commercial of experience in the Australian biotech Eyes Pty Ltd., and Clinical Programme Michael Sistenich, MSc Lawrence Gozlan, BSc (Hons) industry, working with numerous contract Director at Starpharma Holdings Ltd. Director, Chair Remuneration Committee Director, Chair Nomination Committee manufacturing organizations overseas and Formerly held roles at SmithKline Beecham locally in all facets of translational CMC and GlaxoSmithKline with project • Appointed in November 2015 • Appointed in July 2020 from concept through to Phase 2 studies management and clinical operations • Selected Experience: Healthcare specialist in • Selected Experience: Biotechnology investor and responsibilities. international investment management and investment advisor. Life Sciences Investment Manager at Jagen banking. Previously led the Bell Potter team which Pty Ltd, an international private investment advised the Company through a financing in 2014 and organization. Chief Investtment Officer and Founder served as Director of international Equities and Head of of Scientia Capital providing investment advice for Global Healthcare Investments at DWS, Deutsche Bank high net-worth individuals, family offices and Frankfurt. Currently Chairman of Enlitic Inc. Long- institutional investors. Previously responsible for the standing capital market connections and experience in largest biotechnology investment portfolio in Australia the global healthcare investment community. as the institutional biotechnology analyst at the Queensland Investment Corporation, QIC(1) Serves on the Board of Directors4
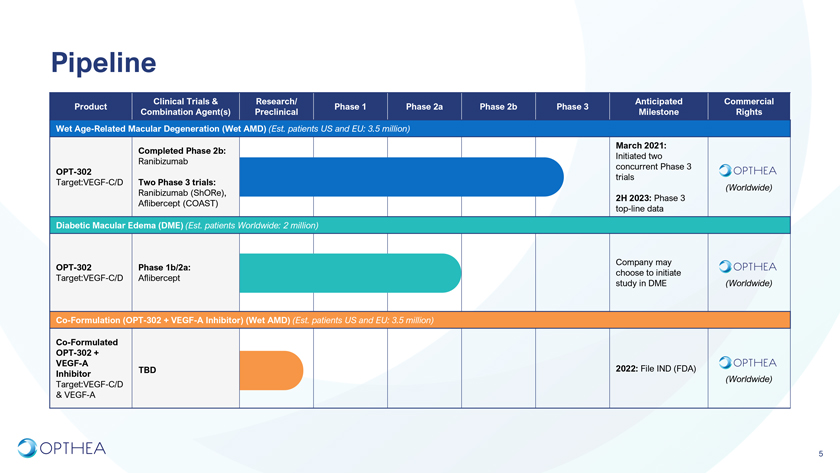
PipelineClinical Trials & Research/ Anticipated Commercial Product Phase 1 Phase 2a Phase 2b Phase 3 Combination Agent(s) Preclinical Milestone Rights Wet Age-Related Macular Degeneration (Wet AMD) (Est. patients US and EU: 3.5 million) March 2021: Completed Phase 2b: Initiated two Ranibizumab concurrent Phase 3 OPT-302 trials Target:VEGF-C/D Two Phase 3 trials: (Worldwide)Ranibizumab (ShORe),2H 2023: Phase 3 Aflibercept (COAST) top-line dataDiabetic Macular Edema (DME) (Est. patients Worldwide: 2 million)Company may OPT-302 Phase 1b/2a: choose to initiate Target:VEGF-C/D Aflibercept study in DME (Worldwide)Co-Formulation (OPT-302 + VEGF-A Inhibitor) (Wet AMD) (Est. patients US and EU: 3.5 million)Co-Formulated OPT-302 + VEGF-A TBD 2022: File IND (FDA)Inhibitor(Worldwide)Target:VEGF-C/D & VEGF-A5
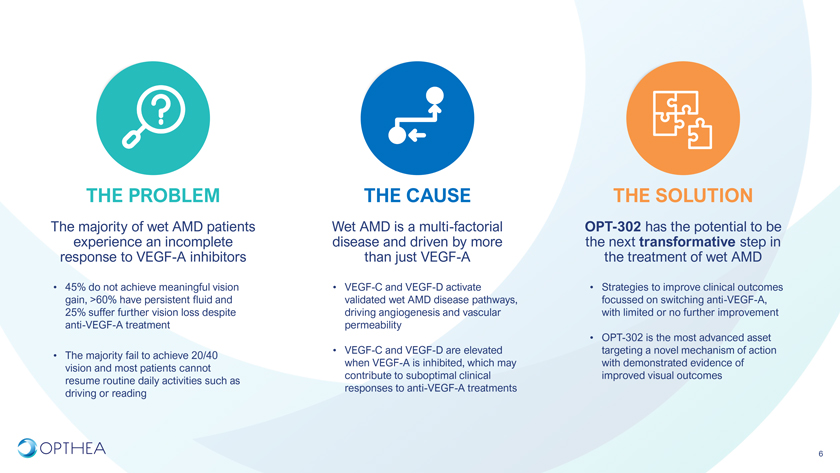
THE PROBLEM THE CAUSE THE SOLUTIONThe majority of wet AMD patients Wet AMD is a multi-factorial OPT-302 has the potential to be experience an incomplete disease and driven by more the next transformative step in response to VEGF-A inhibitors than just VEGF-A the treatment of wet AMD• 45% do not achieve meaningful vision • VEGF-C and VEGF-D activate • Strategies to improve clinical outcomes gain, >60% have persistent fluid and validated wet AMD disease pathways, focussed on switching anti-VEGF-A, 25% suffer further vision loss despite driving angiogenesis and vascular with limited or no further improvement anti-VEGF-A treatment permeability• OPT-302 is the most advanced asset • VEGF-C and VEGF-D are elevated targeting a novel mechanism of action • The majority fail to achieve 20/40 when VEGF-A is inhibited, which may with demonstrated evidence of vision and most patients cannot contribute to suboptimal clinical improved visual outcomes resume routine daily activities such as responses to anti-VEGF-A treatments driving or reading6

Improved EFFICACY is the #1 Unmet Medical Need in Wet AMDImproved initial & sustained visions Improved Efficacy gains and/or lesion morphologyMaintained or improved IMPORTANCE Excellent Safety safety/tolerabilityINCREASED Extended Dosing intervals Less frequent dosing visitsBoulanger-Scemama E, Querques G, About F, Puche, et al. Ranibizumab for exudative age-related macular degeneration: A five year study of adherence to follow-up in a real life setting. J Fr Ophtalmol. 2015 Sep;38(7):620–7.Boyle J, Vukicevic M, Koklanis K, et al. Experiences of patients undergoing repeated intravitreal anti-vascular endothelial growth factor injections for neovascular age-related macular degeneration. Psychol Health Med. 2018 Feb;23(2):127–40.Mueller S, Agostini H, Ehlken C, Bauer-Steinhusen U, Hasanbasic Z, Wilke T. Patient preferences in the treatment of neovascular age-related macular degeneration: A discrete choice experiment. Ophthalmology. 2016 Apr;123(4):876–83. 7 ClearView Commercial Assessment.
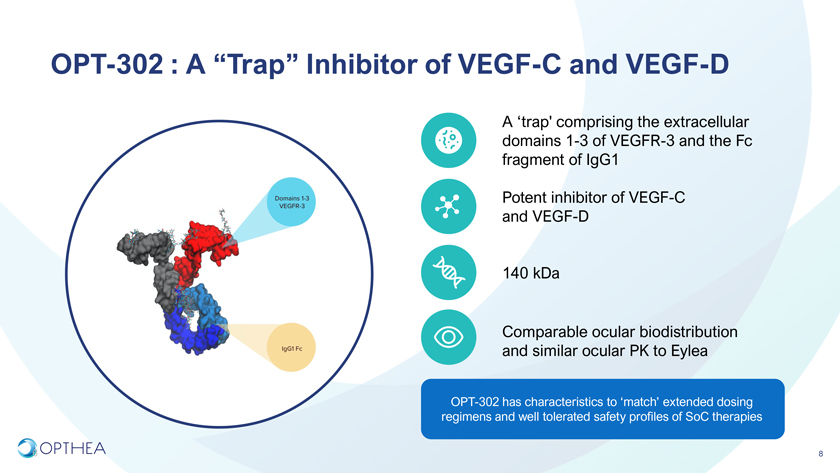
OPT-302 : A “Trap” Inhibitor of VEGF-C and VEGF-DA ‘trap’ comprising the extracellular domains 1-3 of VEGFR-3 and the Fc fragment of IgG1 Potent inhibitor of VEGF-C and VEGF-D 140 kDaComparable ocular biodistribution and similar ocular PK to EyleaOPT-302 has characteristics to ‘match’ extended dosing regimens and well tolerated safety profiles of SoC therapies6 8
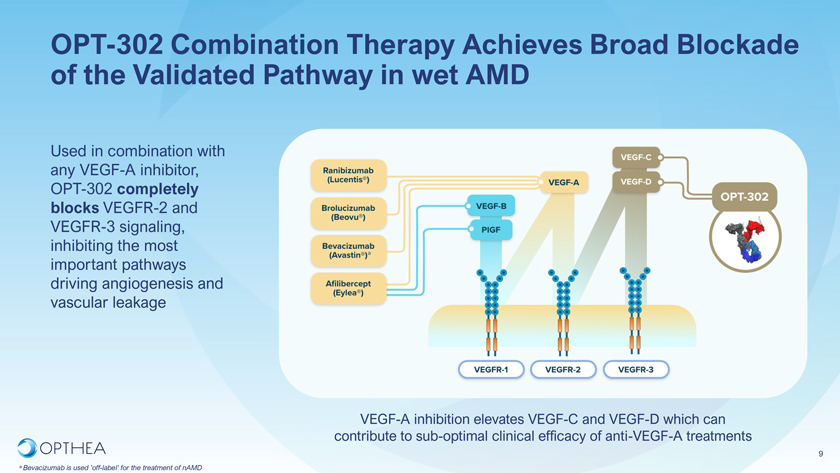
OPT-302 Combination Therapy Achieves Broad Blockade of the Validated Pathway in wet AMDUsed in combination with any VEGF-A inhibitor, OPT-302 completely blocks VEGFR-2 and VEGFR-3 signaling, inhibiting the most important pathways driving angiogenesis and vascular leakage VEGF-A inhibition elevates VEGF-C and VEGF-D which can contribute to sub-optimal clinical efficacy of anti-VEGF-A treatments9
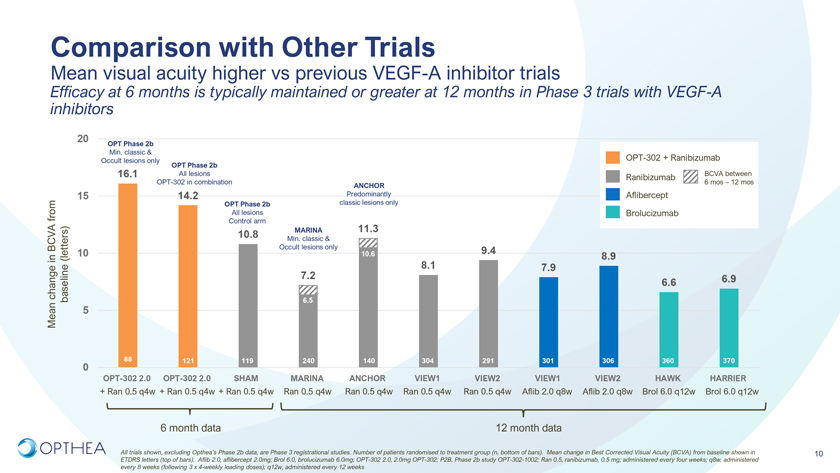
Comparison with Other TrialsMean visual acuity higher vs previous VEGF-A inhibitor trials Efficacy at 6 months is typically maintained or greater at 12 months in Phase 3 trials with VEGF-A inhibitors20OPT Phase 2bMin. classic & Occult lesions only OPT Phase 2b OPT-302 + Ranibizumab16.1 All lesions Ranibizumab BCVA between OPT-302 in combination 6 mos – 12 mosANCHOR15 14.2 Predominantly Aflibercept OPT Phase 2b classic lesions only from All lesions Brolucizumab Control armMARINA 11.310.8 Min. classic & BCVA Occult lesions only 9.4in (letters) 10 10.6 8.9 8.1 7.97.2 6.9 change 6.6 Mean baseline 5 6.588 121 119 240 140 304 291 301 306 360 3700OPT-302 2.0 OPT-302 2.0 SHAM MARINA ANCHOR VIEW1 VIEW2 VIEW1 VIEW2 HAWK HARRIER+ Ran 0.5 q4w + Ran 0.5 q4w + Ran 0.5 q4w Ran 0.5 q4w Ran 0.5 q4w Ran 0.5 q4w Ran 0.5 q4w Aflib 2.0 q8w Aflib 2.0 q8w Brol 6.0 q12w Brol 6.0 q12w6 month data 12 month dataAll trials shown, excluding Opthea’s Phase 2b data, are Phase 3 registrational studies. Number of patients randomised to treatment group (n, bottom of bars). Mean change in Best Corrected Visual Acuity (BCVA) from baseline shown in 10 ETDRS letters (top of bars). Aflib 2.0, aflibercept 2.0mg; Brol 6.0, brolucizumab 6.0mg; OPT-302 2.0, 2.0mg OPT-302; P2B, Phase 2b study OPT-302-1002; Ran 0.5, ranibizumab, 0.5 mg; administered every four weeks; q8w, administered
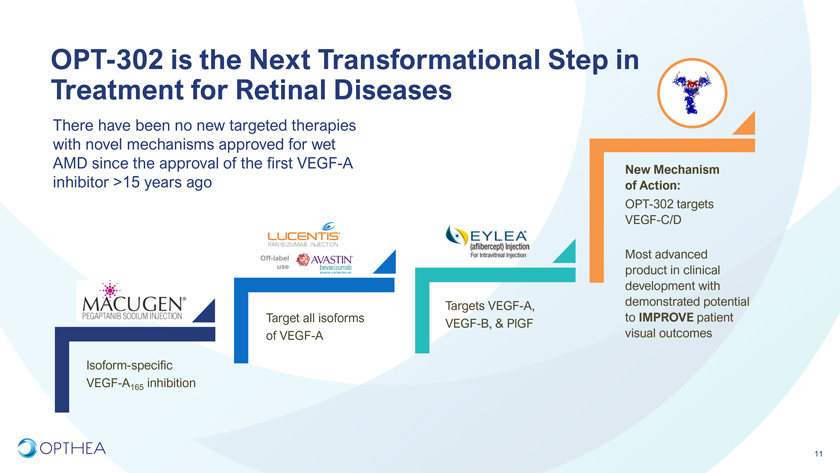
OPT-302 is the Next Transformational Step in Treatment for Retinal DiseasesThere have been no new targeted therapies with novel mechanisms approved for wet AMD since the approval of the first VEGF-A New Mechanism inhibitor >15 years ago of Action:OPT-302 targets VEGF-C/DOff-label Most advanced use product in clinical development with Targets VEGF-A, demonstrated potential Target all isoforms to IMPROVE patient VEGF-B, & PlGF of VEGF-A visual outcomesIsoform-specific VEGF-A165 inhibition11
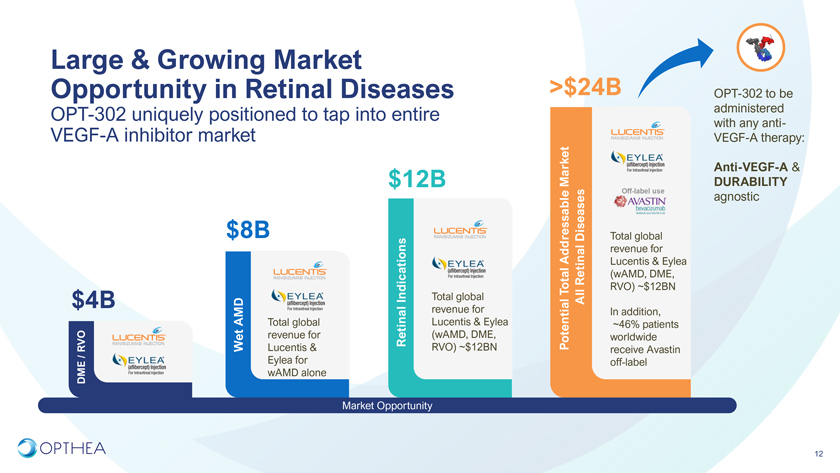
Large & Growing Market Opportunity in Retinal Diseases >$24B OPT-302 to be OPT-302 uniquely positioned to tap into entire administered with any anti-VEGF-A inhibitor market VEGF-A therapy: Anti-VEGF-A & $12B Market DURABILITY Off-label useagnostic$8B Diseases Total global Addressable revenue for Lucentis & Eylea Retinal (wAMD, DME, Total RVO) ~$12BNIndications Total global All$4B revenue for Lucentis & Eylea In addition, AMD Total global ~46% patients revenue for Retinal (wAMD, DME, Potential worldwide RVO Wet Lucentis & RVO) ~$12BN receive Avastin / Eylea for off-label DME wAMD alone Market Opportunity12

Strong OPT-302 Commercial Value Drivers 1 Large growing patient population 2 3 Favorable competitive High unmet need Highly differentiated asset landscape Large market opportunity • 3.5 M wet AMD patients (US & EU) • First and only VEGF-C/D ‘trap’ • No other asset with potential to disrupt treatment • >45% of anti-VEGF A treated patients do not • Only current therapy to have demonstrated paradigm on basis of EFFICACY for wet AMD in achieve meaningful vision gain superior visual outcomes over anti-VEGF–A near or long-term pipeline • $12B global market opportunity therapy • Biosimilars will accelerate OPT-302 uptake 4 5 6 Financially attractive –Expedited regulatory pathway Commercially scalable short & long term • Fast Track designation granted for OPT-302 • 1500 centralized retina specialists in USA enables • Multi-billion dollar peak sales opportunity in USA combination therapy for nAMD lean commercial organization and EU for wet AMD • Offers earlier and expedited review opportunities • 80 -100 sales reps anticipated (<150 total FTEs • Additional multi-billion dollar peak sales across commercial organization) opportunity in DME, RVO, and PCV • Composition of Matter and Methods of Use Patents till 2034 • Further opportunity for Patent Term Extension (PTE), Data and Market Exclusivity periods beyond 2034 13

Strong OPT-302 Commercial Value Drivers1 Large growing patient population 2 3 Favorable competitive High unmet need Highly differentiated asset landscape Large market opportunity• 3.5 M wet AMD patients (US & EU) • First and only VEGF-C/D ‘trap’ • No other asset with potential to disrupt treatment • >45% of anti-VEGF A treated patients do not • Only current therapy to have demonstrated paradigm on basis of EFFICACY for wet AMD in achieve meaningful vision gain superior visual outcomes over anti-VEGF–A near or long-term pipeline• $12B global market opportunity therapy• Biosimilars will accelerate OPT-302 uptake 4 5 6Financially attractive –Expedited regulatory pathway Commercially scalable short & long term• Fast Track designation granted for OPT-302 • 1500 centralized retina specialists in USA enables • Multi-billion dollar peak sales opportunity in USA combination therapy for nAMD lean commercial organization and EU for wet AMD• Offers earlier and expedited review opportunities • 80 -100 sales reps anticipated (<150 total FTEs • Additional multi-billion dollar peak sales across commercial organization) opportunity in DME, RVO, and PCV • Composition of Matter and Methods of Use Patents till 2034• Further opportunity for Patent Term Extension (PTE), Data and Market Exclusivity periods beyond 203413Strong OPT-302 Stakeholder Value DriversPATIENTS RETINA SPECIALISTS PAYERS• Superior gains in visual acuity over • Potential to maximise visual acuity and • Payers want better efficacy outcomes standard of care treatments meaningfully improve long-term outcomes for patients from retinal therapy improve quality of life, independence and • VEGF–A therapy represents a ability to continue routine daily activities • Seamlessly integrates into current anti- significant budget burden to all payers VEGF-A clinical practicesuch as driving and reading • Profound cost inefficiency considering • OPT-302 injected within minutes following limitations of anti-VEGF-A efficacy • Negligible additional treatment burden anti-VEGF-A injection (>45% of anti-VEGF-A paid treatments • Utilizes same diagnostics: OCT and are limited in efficacy) • OPT-302 administered at the same anti- Fluorescein AngiographyVEGF-A injection visit • Payers unlikely to pay for longer • OPT-302 is agnostic to anti-VEGF–A durability or convenience; will reimburse treatment type and duration for patient efficacy and value to the • Advantageous clinic practice financials: healthcare system• No increased staff, visits or diagnostics • Better clinical outcomes represent better • Potential to increase revenue with OPT–302 health economics injections and volume-based discounts • Biosimilars will increase health cost effectiveness for anti-VEGF–A and OPT–302 sequential IVT injections14

Clinical Data:Phase 1/2a trial in treatment naïve and prior treated wet AMD patients (n=51)Phase 2b randomized, controlled, double-masked & statistically powered wet AMD trial (n=366)Phase 1b/2a trial in prior-treated DME patients15
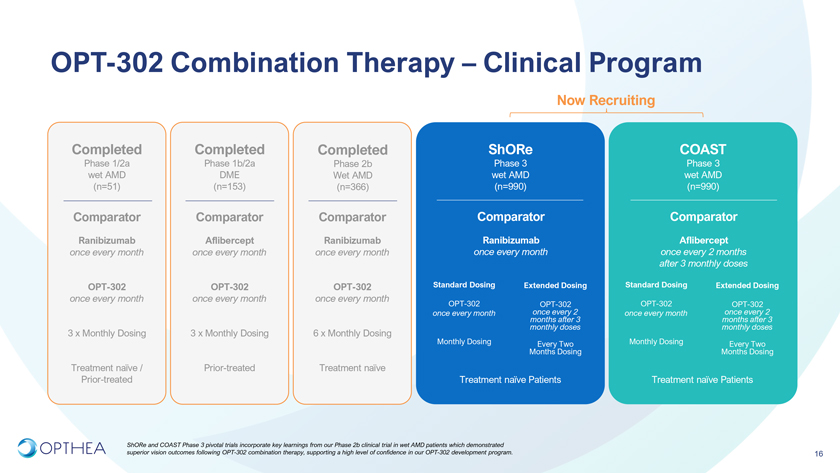
OPT-302 Combination Therapy – Clinical ProgramNow RecruitingCompleted Completed Completed ShORe COASTPhase 1/2a Phase 1b/2a Phase 2b Phase 3 Phase 3 wet AMD DME Wet AMD wet AMD wet AMD (n=51) (n=153) (n=366) (n=990) (n=990)Comparator Comparator Comparator Comparator ComparatorRanibizumab Aflibercept Ranibizumab Ranibizumab Aflibercept once every month once every month once every month once every month once every 2 months after 3 monthly doses OPT-302 OPT-302 OPT-302 Standard Dosing Extended Dosing Standard Dosing Extended Dosingonce every month once every month once every monthOPT-302 OPT-302 OPT-302 OPT-302 once every month months once every after 2 3 once every month months once every after 2 3 monthly doses monthly doses3 x Monthly Dosing 3 x Monthly Dosing 6 x Monthly DosingMonthly Dosing Every Two Monthly Dosing Every Two Months Dosing Months DosingTreatment naïve / Prior-treated Treatment naïve Prior-treated Treatment naïve Patients Treatment naïve Patients ShORe and COAST Phase 3 pivotal trials incorporate key learnings from our Phase 2b clinical trial in wet AMD patients which demonstrated superior vision outcomes following OPT-302 combination therapy, supporting a high level of confidence in our OPT-302 development program. 16

Phase 2bA multicenter, randomized, double-masked, sham controlled study of intravitreal OPT-302 in combination with ranibizumab, in participants with neovascular (wet) AMDConducted at 109 sites across 10 countries: US, EU, Israel OPT-302-1002; NCT ClinicalTrials.gov Identifier: NCT0334508217
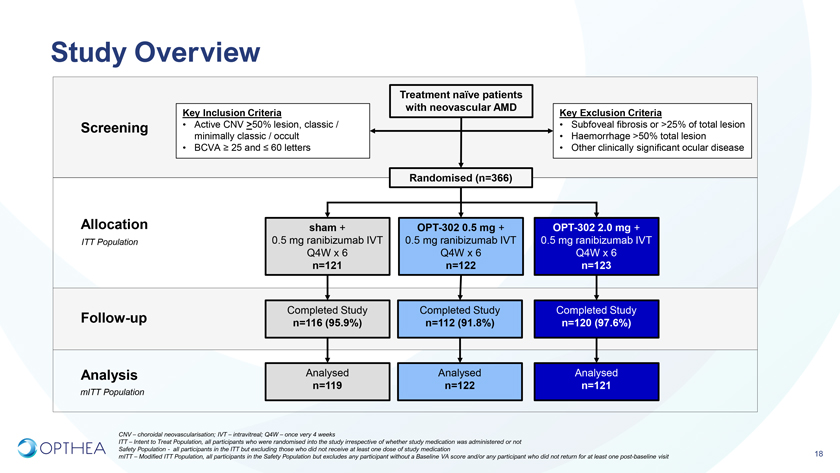
Study OverviewTreatment naïve patients with neovascular AMDKey Inclusion Criteria Key Exclusion Criteria Screening • Active CNV >50% lesion, classic / • Subfoveal fibrosis or >25% of total lesion minimally classic / occult • Haemorrhage >50% total lesion • BCVA ³ 25 and £ 60 letters • Other clinically significant ocular diseaseRandomised (n=366)Allocation sham + OPT-302 0.5 mg + OPT-302 2.0 mg +ITT Population 0.5 mg ranibizumab IVT 0.5 mg ranibizumab IVT 0.5 mg ranibizumab IVT Q4W x 6 Q4W x 6 Q4W x 6 n=121 n=122 n=123Follow-up Completed Study Completed Study Completed Study n=116 (95.9%) n=112 (91.8%) n=120 (97.6%)Analysis Analysed Analysed Analysed n=119 n=122 n=121mITT PopulationCNV – choroidal neovascularisation; IVT – intravitreal; Q4W – once very 4 weeksITT – Intent to Treat Population, all participants who were randomised into the study irrespective of whether study medication was administered or not Safety Population—all participants in the ITT but excluding those who did not receive at least one dose of study medication mITT – Modified ITT Population, all participants in the Safety Population but excludes any participant without a Baseline VA score and/or any participant who did not return for at least one post-baseline visit 18
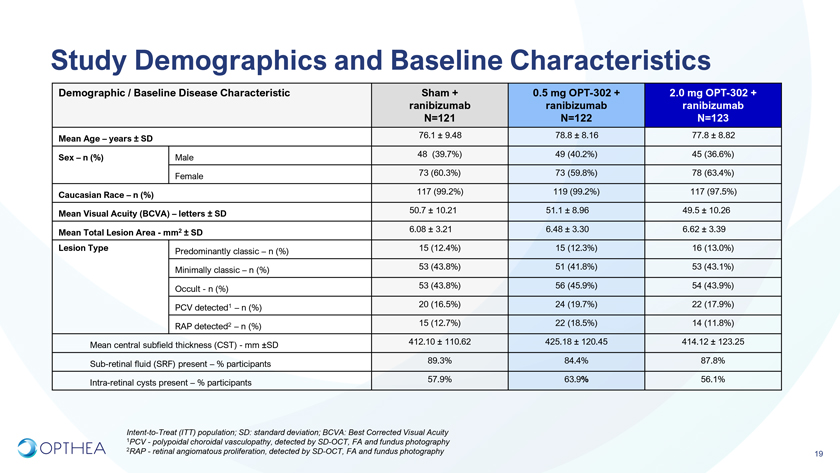
Study Demographics and Baseline CharacteristicsDemographic / Baseline Disease Characteristic Sham + 0.5 mg OPT-302 + 2.0 mg OPT-302 + ranibizumab ranibizumab ranibizumab N=121 N=122 N=123Mean Age – years ± SD 76.1 ± 9.48 78.8 ± 8.16 77.8 ± 8.82 Sex – n (%) Male 48 (39.7%) 49 (40.2%) 45 (36.6%) Female 73 (60.3%) 73 (59.8%) 78 (63.4%) Caucasian Race – n (%) 117 (99.2%) 119 (99.2%) 117 (97.5%)Mean Visual Acuity (BCVA) – letters ± SD 50.7 ± 10.21 51.1 ± 8.96 49.5 ± 10.26 Mean Total Lesion Area—mm2 ± SD 6.08 ± 3.21 6.48 ± 3.30 6.62 ± 3.39Lesion Type Predominantly classic – n (%) 15 (12.4%) 15 (12.3%) 16 (13.0%) Minimally classic – n (%) 53 (43.8%) 51 (41.8%) 53 (43.1%) Occult—n (%) 53 (43.8%) 56 (45.9%) 54 (43.9%) PCV detected1 – n (%) 20 (16.5%) 24 (19.7%) 22 (17.9%) RAP detected2 – n (%) 15 (12.7%) 22 (18.5%) 14 (11.8%) Mean central subfield thickness (CST)—mm ±SD 412.10 ± 110.62 425.18 ± 120.45 414.12 ± 123.25 Sub-retinal fluid (SRF) present – % participants 89.3% 84.4% 87.8% Intra-retinal cysts present – % participants 57.9% 63.9% 56.1%Intent-to-Treat (ITT) population; SD: standard deviation; BCVA: Best Corrected Visual Acuity 1PCV—polypoidal choroidal vasculopathy, detected by SD-OCT, FA and fundus photography 2RAP—retinal angiomatous proliferation, detected by SD-OCT, FA and fundus photography19
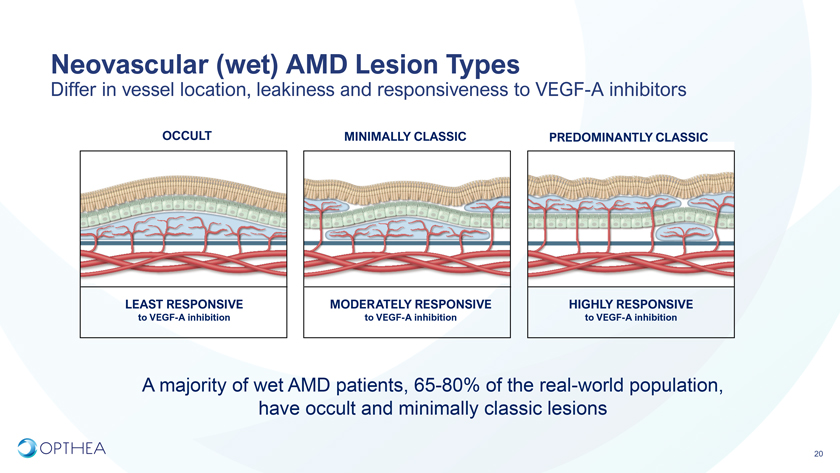
Neovascular (wet) AMD Lesion TypesDiffer in vessel location, leakiness and responsiveness to VEGF-A inhibitorsOCCULT MINIMALLY CLASSIC PREDOMINANTLY CLASSICLEAST RESPONSIVE MODERATELY RESPONSIVE HIGHLY RESPONSIVE to VEGF-A inhibition to VEGF-A inhibition to VEGF-A inhibitionA majority of wet AMD patients, 65-80% of the real-world population, have occult and minimally classic lesions20
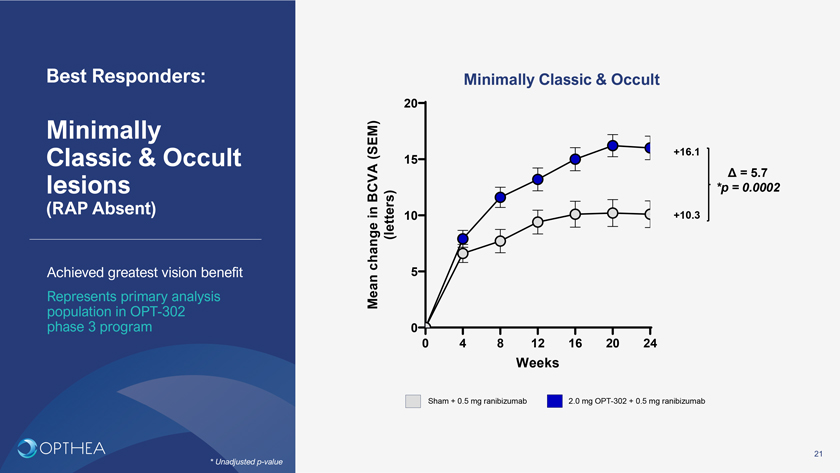
Best Responders: Minimally Classic & Occult20Minimally (SEM) +16.1Classic & Occult 15Ä = 5.7lesions BCVA *p = 0.0002(RAP Absent) in (letters) 10 +10.3 change 5Achieved greatest vision benefitRepresents primary analysis Mean population in OPT-302 phase 3 program 00 4 8 12 16 20 24WeeksSham + 0.5 mg ranibizumab 2.0 mg OPT-302 + 0.5 mg ranibizumab21* Unadjusted p-value
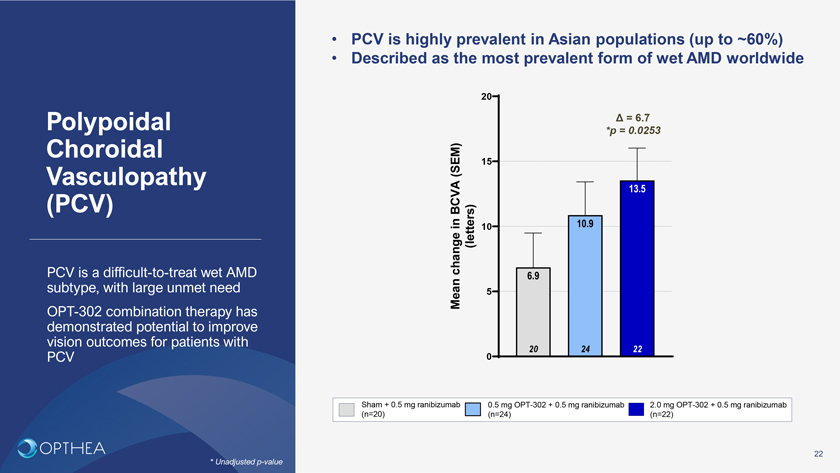
• PCV is highly prevalent in Asian populations (up to ~60%)• Described as the most prevalent form of wet AMD worldwide20Polypoidal Ä = 6.7 Choroidal *p = 0.0253(SEM) 15Vasculopathy 13.5 (PCV) BCVAin 10.910change (letters)PCV is a difficult-to-treat wet AMD 6.9 subtype, with large unmet need Mean 5 OPT-302 combination therapy has demonstrated potential to improve vision outcomes for patients with 20 24 22PCV 0Sham + 0.5 mg ranibizumab 0.5 mg OPT-302 + 0.5 mg ranibizumab 2.0 mg OPT-302 + 0.5 mg ranibizumab (n=20) (n=24) (n=22)22* Unadjusted p-value
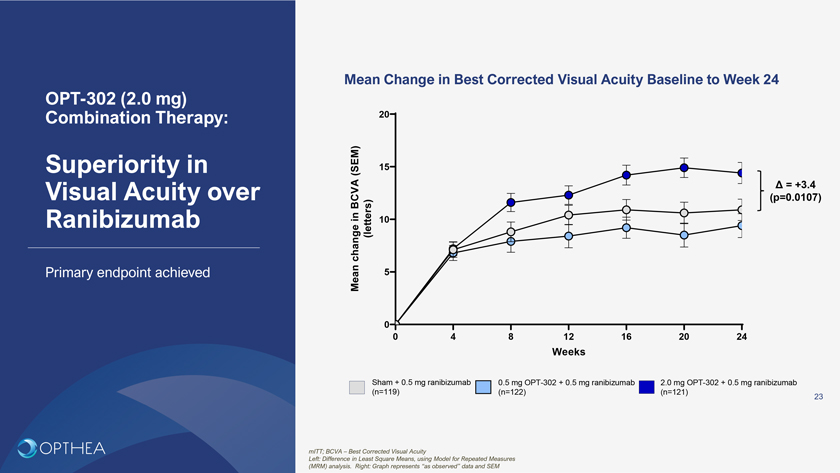
Mean Change in Best Corrected Visual Acuity Baseline to Week 24OPT-302 (2.0 mg) Combination Therapy: 20Superiority in (SEM) 15Visual Acuity over Ä = +3.4 BCVA (p=0.0107) in 10Ranibizumab change (letters)Primary endpoint achieved Mean 500 4 8 12 16 20 24WeeksSham + 0.5 mg ranibizumab 0.5 mg OPT-302 + 0.5 mg ranibizumab 2.0 mg OPT-302 + 0.5 mg ranibizumab (n=119) (n=122) (n=121)23mITT; BCVA – Best Corrected Visual AcuityLeft: Difference in Least Square Means, using Model for Repeated Measures (MRM) analysis. Right: Graph represents “as observed” data and SEM
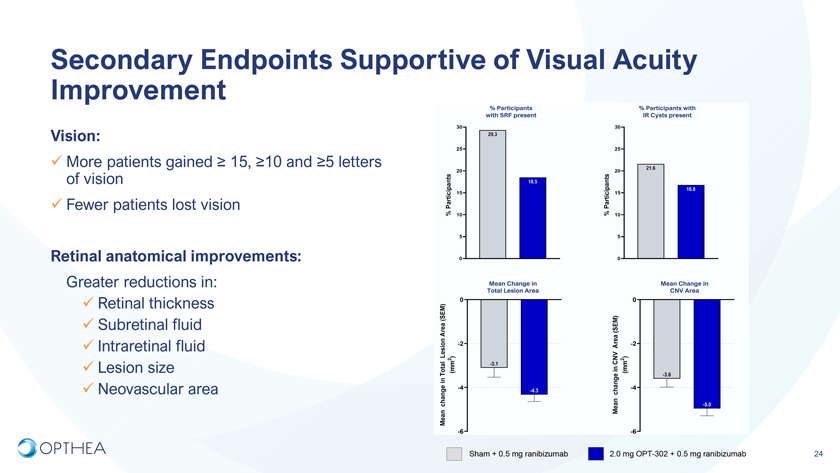
Secondary Endpoints Supportive of Visual Acuity Improvement Vision:More patients gained ³ 15, ³10 and ³5 letters of visionFewer patients lost visionRetinal anatomical improvements:Greater reductions in:Retinal thicknessSubretinal fluidIntraretinal fluidLesion sizeNeovascular areaSham + 0.5 mg ranibizumab 2.0 mg OPT-302 + 0.5 mg ranibizumab 24
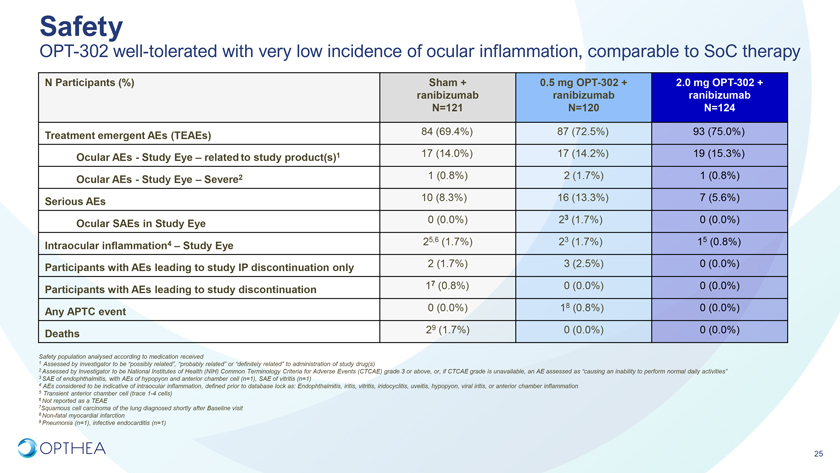
SafetyOPT-302 well-tolerated with very low incidence of ocular inflammation, comparable to SoC therapyN Participants (%) Sham + 0.5 mg OPT-302 + 2.0 mg OPT-302 + ranibizumab ranibizumab ranibizumab N=121 N=120 N=124 Treatment emergent AEs (TEAEs) 84 (69.4%) 87 (72.5%) 93 (75.0%)Ocular AEs—Study Eye – related to study product(s)1 17 (14.0%) 17 (14.2%) 19 (15.3%) Ocular AEs—Study Eye – Severe2 1 (0.8%) 2 (1.7%) 1 (0.8%)Serious AEs 10 (8.3%) 16 (13.3%) 7 (5.6%) Ocular SAEs in Study Eye 0 (0.0%) 23 (1.7%) 0 (0.0%)25,6 (1.7%) 23 (1.7%) 15 (0.8%)Intraocular inflammation4 – Study Eye Participants with AEs leading to study IP discontinuation only 2 (1.7%) 3 (2.5%) 0 (0.0%) Participants with AEs leading to study discontinuation 17 (0.8%) 0 (0.0%) 0 (0.0%)Any APTC event 0 (0.0%) 18 (0.8%) 0 (0.0%) Deaths 29 (1.7%) 0 (0.0%) 0 (0.0%)Safety population analysed according to medication received1 Assessed by investigator to be “possibly related”, “probably related” or “definitely related” to administration of study drug(s)2 Assessed by Investigator to be National Institutes of Health (NIH) Common Terminology Criteria for Adverse Events (CTCAE) grade 3 or above, or, if CTCAE grade is unavailable, an AE assessed as “causing an inability to perform normal daily activities”3 SAE of endophthalmitis, with AEs of hypopyon and anterior chamber cell (n=1), SAE of vitritis (n=1)4 AEs considered to be indicative of intraocular inflammation, defined prior to database lock as: Endophthalmitis, iritis, vitritis, iridocyclitis, uveitis, hypopyon, viral iritis, or anterior chamber inflammation5 Transient anterior chamber cell (trace 1-4 cells)6 Not reported as a TEAE7Squamous cell carcinoma of the lung diagnosed shortly after Baseline visit8 Non-fatal myocardial infarction9 Pneumonia (n=1), infective endocarditis (n=1)25

Phase 2b trial met primary endpoint OPT-302 (2.0 mg) combination therapy demonstrated superiority in visual acuity over ranibizumab + sham Conclusions Additional +5.7 letter gain observed in high responder subgroup (minimally classic & occult lesions, RAP absent, >70% study population), represents Phase 3 primary analysis populationOPT-302 Phase 2b  Additional +6.7 letter gain (p=0.025)* in PCV lesions, a difficult-to-wet AMD Trial treat wet AMD subtype, predominant in Asian populations, with large unmet need Primary endpoint achieved Additional +3.4 letter gain (p=0.0107) in total patient population High performing ranibizumab control armSecondary outcomes were supportive of the primary endpoint Greater vision gains observed in minimally classic/occult lesions Favorable tolerability profile similar to ranibizumab alone Promising treatment option for wet AMD suitable for Phase 326

Phase 3 Pivotal Trials Two concurrent, randomized, controlled, 3-arm Phase 3 studies investigating OPT-302 administered every four-weeks and every-eight weeks in combination with standard of care anti-VEGF-A therapy:ShORe: Study of OPT-302 in combination with Ranibizumab (Study OPT-302-1004)COAST: Combination OPT-302 with Aflibercept STudy (Study OPT-302-1005)27
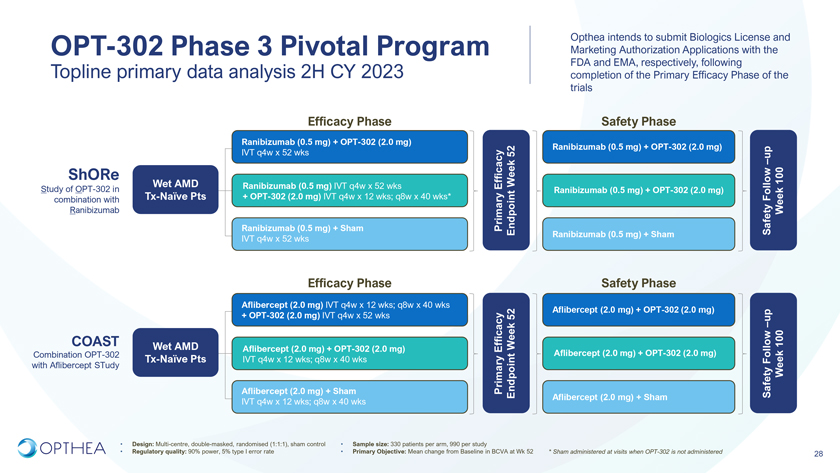
Opthea intends to submit Biologics License and OPT-302 Phase 3 Pivotal Program Marketing Authorization Applications with the Topline primary data analysis 2H CY 2023 FDA and EMA, respectively, following completion of the Primary Efficacy Phase of the trialsEfficacy Phase Safety PhaseRanibizumab (0.5 mg) + OPT-302 (2.0 mg) IVT q4w x 52 wks 52 Ranibizumab (0.5 mg) + OPT-302 (2.0 mg) up–ShORe 100Wet AMD Ranibizumab (0.5 mg) IVT q4w x 52 wks EfficacyWeek Study of OPT-302 in Ranibizumab (0.5 mg) + OPT-302 (2.0 mg) combination with Tx-Naïve Pts + OPT-302 (2.0 mg) IVT q4w x 12 wks; q8w x 40 wks* Follow Ranibizumab WeekRanibizumab (0.5 mg) + Sham PrimaryEndpoint Safety Ranibizumab (0.5 mg) + ShamIVT q4w x 52 wksEfficacy Phase Safety PhaseAflibercept (2.0 mg) IVT q4w x 12 wks; q8w x 40 wks+ OPT-302 (2.0 mg) IVT q4w x 52 wks 52 Aflibercept (2.0 mg) + OPT-302 (2.0 mg) up–COAST Wet AMD 100Aflibercept (2.0 mg) + OPT-302 (2.0 mg) EfficacyWeek Combination OPT-302 Aflibercept (2.0 mg) + OPT-302 (2.0 mg)Tx-Naïve Pts IVT q4w x 12 wks; q8w x 40 wks Follow with Aflibercept STudy WeekAflibercept (2.0 mg) + Sham PrimaryEndpoint SafetyAflibercept (2.0 mg) + ShamIVT q4w x 12 wks; q8w x 40 wks• Design: Multi-centre, double-masked, randomised (1:1:1), sham control • Sample size: 330 patients per arm, 990 per study• Regulatory quality: 90% power, 5% type I error rate • Primary Objective: Mean change from Baseline in BCVA at Wk 52 * Sham administered at visits when OPT-302 is not administered 28

Opthea’s Phase 3 Program is Optimised for SuccessWe know the patients which respond best OPT-302 positioned for use in combination Patients with minimally classic with any VEGF-A inhibitor• & occult lesions are the majority (~80%) of the wet AMD populationInvestigates OPT-302 standard and extended Primary Analysis will be first conducted in the dosing regimens: Minimally Classic/Occult population• Analysis of efficacy on q4w and q8w dosing regimens provides insight into OPT-302 durability• Maximises opportunity to demonstrate most compelling vision benefit• In Phase 2b, +5.7 letters superior benefit of OPT-302 combination therapy compared to Lucentis® alone Meetings to discuss Phase 3 design and analysis plan completed with US (FDA) and Our Phase 3 trials are designed to maximise the European (EMA) regulatory agencies commercial opportunity• Efficacy assessed in minimally classic/occult, followed by ‘all-comer’ total patient population• OPT-302 investigated in combination with two standard of care treatments respectively29

OPT-302: The potential to revolutionize the treatment of wet AMD and improve and preserve vision1 • Large treated (80%) wet AMD market (2.7M patients#) in a US$12B dollar anti-VEGF-A category2Market Background• Standard of Care is anti–VEGF-A injections once per month or every two months by intravitreal delivery• Highest unmet need in wet AMD is EFFICACY, to improve visual outcomes2 • >45% do not achieve meaningful vision gain, >60% have persistent fluid and 25% suffer further vision loss despite anti-High Unmet NeedVEGF-A treatment• Current innovation is focused on durability rather than improving visual outcomes• When combined with anti-VEGF–A, OPT-302 broadly shuts down the VEGF/VEGFR pathways driving angiogenesis and vascular leakage• Only current therapy demonstrating superior visual outcomes over anti-VEGF–A, with comparable safety3 OPT-302: first in class • Phase 2b results demonstrate visual acuity gains over standard of care:VEGF-C/D ‘trap’ • Additional +5.7 letter gain (p=0.0002)* in minimally classic and occult lesion patients (80% of patient population)• Additional +6.7 letter gain (p=0.025)* in PCV lesions, a difficult-to-treat wet AMD subtype, predominant in Asian populations, with large unmet need• Additional +3.4 letter gain (p=0.0107) in total patient population • FDA Fast Track status granted based on Phase 2b results • Two global pivotal Phase 3 trials, ShORe & COAST, currently recruiting, topline data second half calendar year 20234 • USA launch 2024; EU, Japan, and ROW to followLaunch • Compelling patient, physician and payer value propositions will propel acceptance, adoption and uptake• Only VEGF-C/D ‘trap’, no viable threat in competitive pipelines5 • Multi-billion dollar commercial opportunity in USA and in EU for wet AMD aloneFinancials • Additional indications DME, RVO, polypoidal (PCV) wet AMD represent blockbuster upside opportunity• Strong Composition of Matter and Methods of Use patents till 2034• Further opportunity for Patent Term Extension (PTE), Data and Market Exclusivity periods beyond 203430# USA & EU; * Unadjusted p-value

Megan Baldwin, PhD CEO & Managing Director Opthea Limited Level 4, 650 Chapel Street South Yarra 3141 Victoria Australia P: 788 +61 674 9826 E: 0399 M: +61 447 megan.baldwin@opthea.com31
Exhibit 99.3

19 October 2021
Market Announcements Office
Australian Securities Exchange Limited
20 Bridge Street SYDNEY NSW 2000
Dear Ms,
Results of 2021 Annual General Meeting
Opthea Limited (ASX: OPT)
In accordance with Listing Rule 3.13.2 and section 251AA of the Corporations Act, we advise details of the resolutions and the proxies received in respect of each resolution are set out in the attached proxy summary.
Yours faithfully

| Karen Adams |
| Company Secretary |
Opthea Limited, Suite 0403, Level 4, 650 Chapel Street, South Yarra, Victoria 3141, Australia
A.C.N. 006 340 567 l Telephone: +61 3 9826 0399 l Website: www.opthea.com
OPTHEA LIMITED
2021 Annual General Meeting
Tuesday, 19 October 2021
Results of Meeting
The following information is provided in accordance with section 251AA(2) of the Corporations Act 2001 (Cth) and ASX Listing Rule 3.13.2
| Resolution details |
Instructions given to validly appointed proxies (as at proxy close) |
Number of votes cast on the poll (where applicable) |
Resolution Result |
If s250U applies | ||||||||||||||||
| Resolution |
Resolution Type |
For | Against | Proxy’s Discretion |
Abstain | For | Against | Abstain* | Carried / Not Carried |
|||||||||||
| 1 Re-Election of Director - Mr Michael Sistenich |
Ordinary |
156,027,253 97.63% |
725,640 0.45% |
3,066,982 1.92% |
314,457 |
159,220,554 99.54% |
738,726 0.46% |
314,457 |
Carried |
|||||||||||
| 2 Re-Election of Director - Mr Lawrence Gozlan |
Ordinary |
156,038,526 97.55% |
870,140 0.54% |
3,051,982 1.91% |
173,684 |
159,216,827 99.45% |
883,226 0.55% |
173,684 |
Carried |
|||||||||||
| 3 Election of Director - Dr Julia Haller |
Ordinary |
156,577,488 97.97% |
193,672 0.12% |
3,057,556 1.91% |
305,616 |
159,764,913 99.87% |
203,208 0.13% |
305,616 |
Carried |
|||||||||||
| 4 Election of Director - Ms Judith Robertson |
Ordinary |
156,389,577 97.85% |
388,882 0.24% |
3,051,982 1.91% |
303,891 |
159,571,428 99.75% |
398,418 0.25% |
303,891 |
Carried |
|||||||||||
| 5 Adoption of remuneration report |
Ordinary |
145,985,457 96.39% |
3,806,889 2.51% |
1,659,345 1.10% |
3,414,406 |
147,763,121 97.48% |
3,819,975 2.52% |
3,414,406 |
Carried |
|||||||||||
| 6 Approval and adoption of amended and restated long-term incentive plan | Ordinary |
155,805,163 97.38% |
1,089,864 0.68% |
3,105,221 1.94% |
134,084 |
159,032,253 99.31% |
1,099,400 0.69% |
134,084 |
Carried |
|||||||||||
| 7 Approval of issue of performance rights awarded to rights awarded to Dr Megan Baldwin | Ordinary |
122,475,564 78.41% |
30,644,239 19.62% |
3,081,293 1.97% |
93,838 |
125,678,726 80.39% |
30,653,775 19.61% |
93,838 |
Carried |
|||||||||||
| 8 Issue of options to Dr Julia Haller under the nonexecutive director share and option plan |
Ordinary |
124,217,601 77.65% |
32,681,359 20.42% |
3,101,370 1.93% |
134,002 |
127,440,158 79.58% |
32,691,577 20.42% |
134,002 |
Carried |
|||||||||||
| 9 Issue of options to Ms Judith Robertson under the non-executive director share and option plan | Ordinary |
124,209,563 77.64% |
32,689,841 20.43% |
3,100,970 1.93% |
133,958 |
127,431,720 79.58% |
32,700,059 20.42% |
133,958 |
Carried |
|||||||||||
| 10 Election of Director - Dr Jeremy Levin |
Ordinary |
122,695,684 77.98% |
31,683,412 20.14% |
2,955,975 1.88% |
571,226 |
125,769,978 79.87% |
31,696,498 20.13% |
571,226 |
Carried |
|||||||||||
| * | Votes cast by a person who abstains on an item are not counted in calculating the required majority on a poll. |
Exhibit 99.4

ASX and Media Release
19 October 2021
Opthea Opens Patient Enrollment in the Asia-Pacific Region for the
OPT-302 ShORe and COAST Phase 3 Trials in Wet AMD
Melbourne, Australia; 19 October 2021 – Opthea Limited (ASX:OPT; Nasdaq:OPT), a clinical stage biopharmaceutical company developing novel therapies to treat highly prevalent and progressive retinal diseases, today announced that the first clinical trial sites in the Asia-Pacific region are now open for patient enrollment into the OPT-302 Phase 3 pivotal clinical program for the treatment of wet (neovascular) age-related macular degeneration (AMD). The clinical trial sites, located in Australia, further build upon the progress of other international regions including the U.S., Canada and Europe that are also actively recruiting patients.
“We are delighted to open enrollment in the Asia-Pacific region. This is a significant milestone for the OPT-302 development program and enables eligible patients to participate in the ShORe and COAST Phase 3 trials which are designed based on strong scientific rationale, prior positive clinical efficacy results and extensive safety data in the wet AMD target population. Over the following weeks we anticipate opening sites in additional countries in the Asia Pacific region including South Korea and the Philippines,” said Dr Megan Baldwin, CEO and Managing Director of Opthea.
The ShORe and COAST studies are both double-masked, sham-controlled Phase 3 registrational trials to evaluate efficacy and safety of intravitreal 2.0 mg OPT-302 in combination with either 0.5 mg ranibizumab (Lucentis®), or 2.0 mg aflibercept (Eylea®) respectively. The primary endpoint of the studies is the mean change in best corrected visual acuity from baseline to week 52 for OPT-302 combination therapy compared to standard of care anti-VEGF-A monotherapy. The read-out of the top-line results through 52 weeks is anticipated in the second half of 2023, and following completion of this primary efficacy phase, the Company plans to file Biologics License and Marketing Authorization Applications with the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) respectively.
Recently, the FDA granted Fast Track status for OPT-302, in combination with anti-VEGF-A therapy for the treatment of patients with wet AMD and this regulatory designation offers benefits to help advance development and expedite the review of novel therapies for serious conditions for which there is an unmet medical need, with the aim of getting important new therapies to patients more quickly.
Additional information on Opthea’s technology and the Phase 3 pivotal clinical trials can be found at www.opthea.com and at ClinicalTrials.gov (ShORe trial, ID#: NCT04757610; COAST trial, ID#: NCT04757636).
Level 4, 650 Chapel Street, South Yarra, Victoria 3141 Australia T +61 (3) 9826 0399 F +61 (3) 9824 0083
www.opthea.com ABN 32 006 340 567
About Opthea
Opthea (ASX:OPT; Nasdaq:OPT) is a biopharmaceutical company developing novel therapies to address the unmet need in the treatment of highly prevalent and progressive retinal diseases, including wet age-related macular degeneration (wet AMD) and diabetic macular edema (DME). Opthea’s lead product candidate OPT-302 is in pivotal Phase 3 clinical trials and being developed for use in combination with anti-VEGF-A monotherapies to achieve broader inhibition of the VEGF family, with the goal of improving overall efficacy and demonstrating superior vision gains over that which can be achieved by inhibiting VEGF-A alone.
Inherent risks of Investment in Biotechnology Companies
There are a number of inherent risks associated with the development of pharmaceutical products to a marketable stage. The lengthy clinical trial process is designed to assess the safety and efficacy of a drug prior to commercialization and a significant proportion of drugs fail one or both of these criteria. Other risks include uncertainty of patent protection and proprietary rights, whether patent applications and issued patents will offer adequate protection to enable product development, the obtaining of necessary drug regulatory authority approvals and difficulties caused by the rapid advancements in technology. Companies such as Opthea are dependent on the success of their research and development projects and on the ability to attract funding to support these activities. Investment in research and development projects cannot be assessed on the same fundamentals as trading and manufacturing enterprises. Therefore, investment in companies specializing in drug development must be regarded as highly speculative. Opthea strongly recommends that professional investment advice be sought prior to such investments.
Forward-looking statements
Certain statements in this announcement may contain forward-looking statements, including within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. Any statement describing Company goals, expectations, intentions or beliefs is a forward-looking statement and should be considered an at risk statement, including, but not limited to, the continuation of patient recruitment for Opthea’s pivotal Phase 3 clinical trials of OPT-302 in wet AMD. Such statements are based on Opthea’s current plans, objectives, estimates, expectations and intentions and are subject to certain risks and uncertainties, including risks and uncertainties associated with clinical trials and product development and the impact of general economic, industry or political conditions in Australia, the United States or internationally. These and other risks and uncertainties are described more fully in the section titled “Risk Factors” in the final prospectus filed with the SEC on October 19, 2020. The Company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events, or otherwise, except as required under applicable law. You should not place undue reliance on these forward-looking statements as predictions of future events, which statements apply only as of the date of this announcement. Actual results could differ materially from those discussed in this ASX announcement.
Authorized for release to ASX by Megan Baldwin, CEO & Managing Director
|
Company & Media Enquiries: |
Join our email database to receive program updates: | |
| U.S.A. & International: Sam Martin Argot Partners Tel: +1 212-600-1902 opthea@argotpartners.com
Australia: Rudi Michelson Monsoon Communications Tel: +61 (0) 3 9620 3333 |
Tel: +61 (0) 3 9826 0399 info@opthea.com www.opthea.com | |
Level 4, 650 Chapel Street, South Yarra, Victoria 3141 Australia T +61 (3) 9826 0399 F +61 (3) 9824 0083
www.opthea.com ABN 32 006 340 567
