6-K
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
OF THE SECURITIES EXCHANGE ACT OF 1934
For the month of August 2025
Commission File No. 001-39621
OPTHEA LIMITED
(Translation of registrant’s name into English)
Level 4
650 Chapel Street
South Yarra, Victoria, 3141
Australia
(Address of registrant’s principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereto duly authorized.
|
|
|
|
OPTHEA LIMITED |
|
(Registrant) |
|
|
|
|
By: |
/s/ Frederic Guerard |
|
Name: |
Frederic Guerard |
|
Title: |
Chief Executive Officer |
Date: 08/20/2025

CORPORATE UPDATE�INVESTOR CALL – 20 August 2025 Dr Jeremy Levin – Chairman of the Board Fred Guerard – Chief Executive Officer Tom Reilly – Chief Financial Officer

This investor presentation (Presentation) is dated August 19th, 2025 and has been prepared by Opthea Limited (ASX:OTP) (Opthea or the Company). SUMMARY INFORMATION This Presentation contains summary information about the current activities of Opthea and its subsidiaries (the Opthea Group or Group) which is current as at the date of this Presentation unless otherwise indicated. The information in this Presentation is of a general nature and does not purport to be complete. This Presentation does not purport to contain all of the information that an investor should consider when making an investment decision. It should be read in conjunction with Opthea’s other periodic and continuous disclosure announcements, available from the ASX at www.asx.com.au. Certain market and industry data used in this Presentation may have been obtained from research, surveys or studies conducted by third parties, including industry or general publications. None of the Opthea Group nor its advisers or representatives have independently verified any such market or industry data provided by third parties or industry or general publications. FORWARD LOOKING STATEMENTS This Presentation contains forward-looking statements which are identified by words such as ‘may’, ‘could’, ‘believes’, ‘estimates’, ‘targets’, ‘expects’, or ‘intends’ and other similar words that involve risks and uncertainties. These statements are based on an assessment of present economic and operating conditions, and on a number of assumptions regarding future events and actions that, as at the date of this Presentation, are considered reasonable. Such forward-looking statements are not a guarantee of future performance and involve known and unknown risks, uncertainties, assumptions and other important factors, many of which are beyond the control of Opthea, the Directors and the management. The Directors cannot and do not give any assurance that the results, performance or achievements expressed or implied by the forward-looking statements contained in this Presentation will actually occur and investors are cautioned not to place undue reliance on these forward-looking statements. The Directors have no intention to update or revise forward-looking statements, or to publish prospective financial information in the future, regardless of whether new information, future events or any other factors affect the information contained in this Presentation, except where required by law or the ASX listing rules. NO FINANCIAL PRODUCT ADVICE This Presentation is for information purposes only and is not a prospectus, disclosure document, product disclosure statement or other offering document under Australian law or the law of any other jurisdiction. This Presentation is not financial product advice or investment advice nor a recommendation to acquire securities and has been prepared without taking into account the objectives, financial situation and particular needs of individuals. Before making any investment decision, prospective investors should consider the appropriateness of the information having regard to their own objectives, financial situation and needs and seek appropriate advice, including financial, legal and taxation advice appropriate to their jurisdiction. Opthea Group is not licenced to provide financial product advice in respect of securities. Important Information

Introduction – Dr Jeremy Levin, Chairman of the Board Results of Sozinibercept Phase 3 Trials (COAST and ShORe) – Fred Guerard, CEO Update on Development Funding Agreement Negotiations – Tom Reilly, CFO Current state of the company – Dr Jeremy Levin, Chairman of the Board The path forward – Dr Jeremy Levin, Chairman of the Board Q&A AGENDA – CORPORATE UPDATE

Introduction��Dr Jeremy Levin

The results of our Phase 3 program did not meet their primary endpoints and were deeply disappointing Thanks to our CEO’s and CFO's efforts, we have now completed negotiations with the Development Funding Agreement investors, gaining clarity on available resources and outcomes for shareholders The purpose of this presentation is to share clinical results with shareholders, provide an update on the DFA outcomes and outline potential opportunities for the company moving forward While there have been challenges, this marks a new chapter for the company INTRODUCTION

Results of Sozinibercept Phase 3 Trials in Participants with Neovascular Age-related Macular Degeneration: COAST (Sozinibercept in Combination with Aflibercept) and ShORe (Sozinibercept in Combination with Ranibizumab)��Fred Guerard
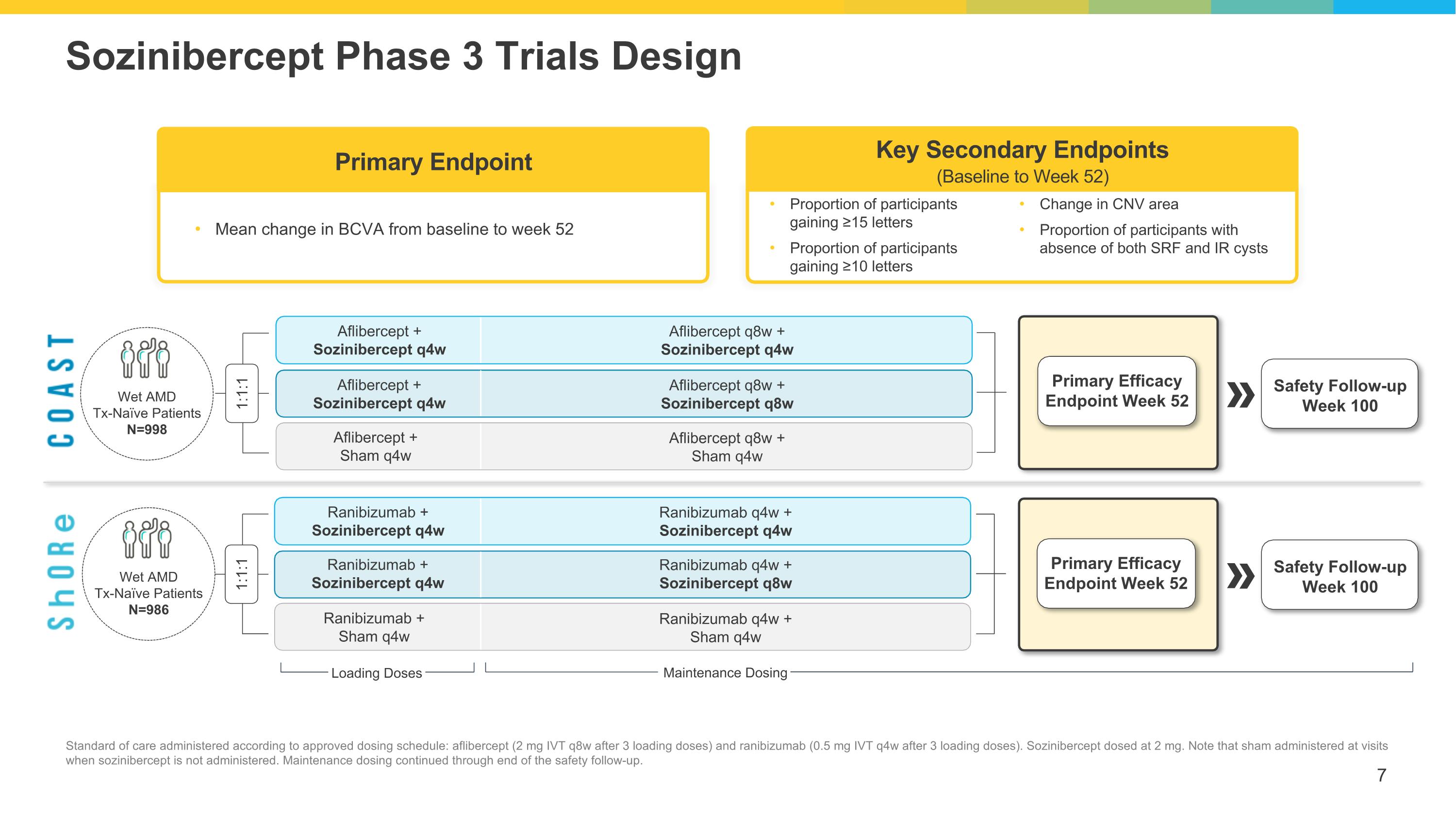
Primary Efficacy Endpoint Week 52 Primary Efficacy Endpoint Week 52 Safety Follow-up Week 100 Safety Follow-up Week 100 Sozinibercept Phase 3 Trials Design Standard of care administered according to approved dosing schedule: aflibercept (2 mg IVT q8w after 3 loading doses) and ranibizumab (0.5 mg IVT q4w after 3 loading doses). Sozinibercept dosed at 2 mg. Note that sham administered at visits when sozinibercept is not administered. Maintenance dosing continued through end of the safety follow-up. Wet AMD Tx-Naïve Patients N=998 Wet AMD Tx-Naïve Patients N=986 1:1:1 1:1:1 Loading Doses Maintenance Dosing Aflibercept q8w + Sozinibercept q4w Aflibercept q8w + Sham q4w Aflibercept q8w + Sozinibercept q8w Aflibercept + Sozinibercept q4w Aflibercept + Sozinibercept q4w Aflibercept + Sham q4w Ranibizumab q4w + Sozinibercept q4w Ranibizumab q4w + Sham q4w Ranibizumab q4w + Sozinibercept q8w Ranibizumab + Sozinibercept q4w Ranibizumab + Sozinibercept q4w Ranibizumab + Sham q4w Mean change in BCVA from baseline to week 52 Primary Endpoint Key Secondary Endpoints (Baseline to Week 52) Proportion of participants gaining ≥15 letters Proportion of participants gaining ≥10 letters Change in CNV area Proportion of participants with absence of both SRF and IR cysts
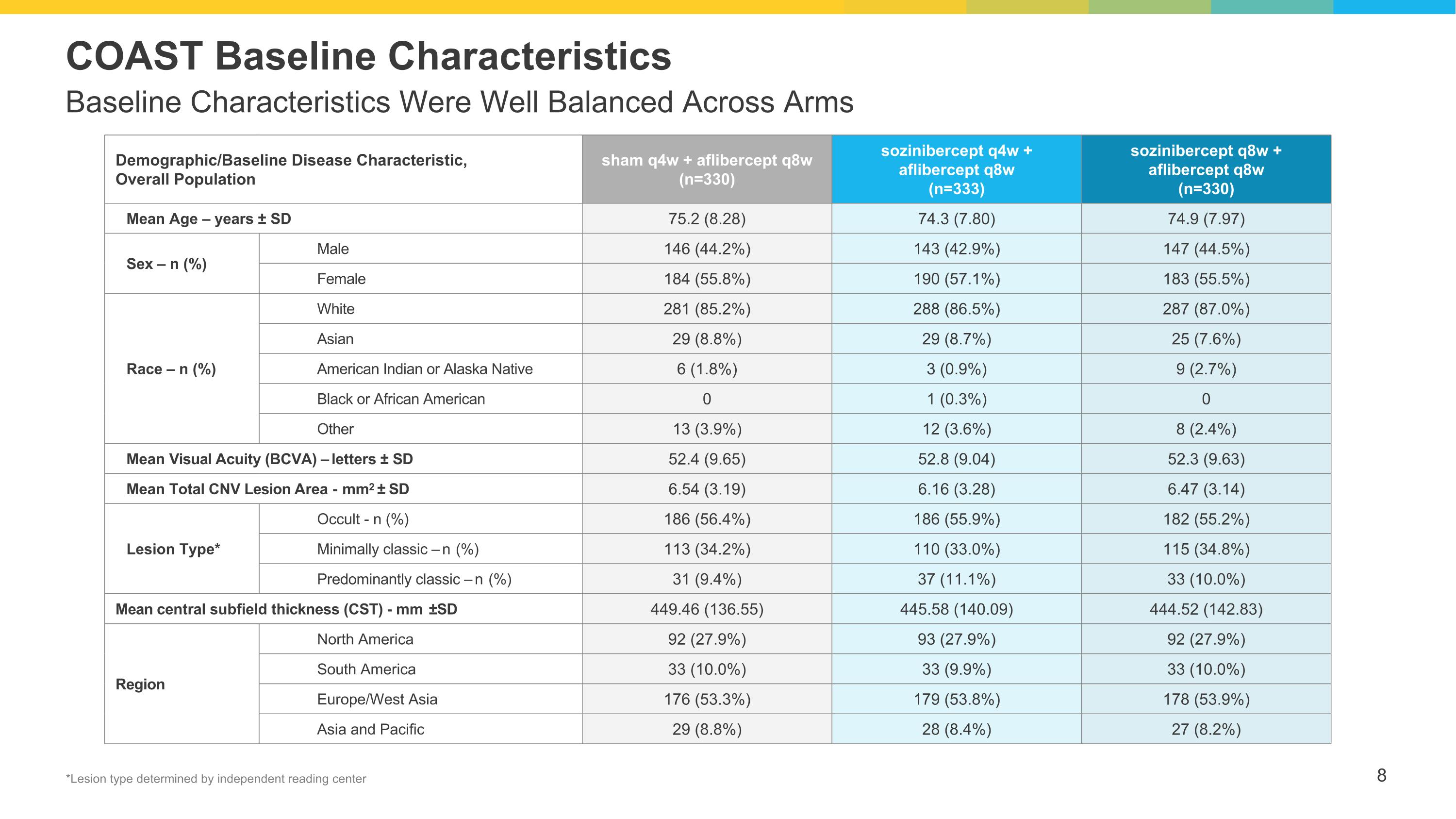
Baseline Characteristics Were Well Balanced Across Arms COAST Baseline Characteristics Demographic/Baseline Disease Characteristic, Overall Population sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) Mean Age – years ± SD 75.2 (8.28) 74.3 (7.80) 74.9 (7.97) Sex – n (%) Male 146 (44.2%) 143 (42.9%) 147 (44.5%) Female 184 (55.8%) 190 (57.1%) 183 (55.5%) Race – n (%) White 281 (85.2%) 288 (86.5%) 287 (87.0%) Asian 29 (8.8%) 29 (8.7%) 25 (7.6%) American Indian or Alaska Native 6 (1.8%) 3 (0.9%) 9 (2.7%) Black or African American 0 1 (0.3%) 0 Other 13 (3.9%) 12 (3.6%) 8 (2.4%) Mean Visual Acuity (BCVA) – letters ± SD 52.4 (9.65) 52.8 (9.04) 52.3 (9.63) Mean Total CNV Lesion Area - mm2 ± SD 6.54 (3.19) 6.16 (3.28) 6.47 (3.14) Lesion Type* Occult - n (%) 186 (56.4%) 186 (55.9%) 182 (55.2%) Minimally classic – n (%) 113 (34.2%) 110 (33.0%) 115 (34.8%) Predominantly classic – n (%) 31 (9.4%) 37 (11.1%) 33 (10.0%) Mean central subfield thickness (CST) - mm ±SD 449.46 (136.55) 445.58 (140.09) 444.52 (142.83) Region North America 92 (27.9%) 93 (27.9%) 92 (27.9%) South America 33 (10.0%) 33 (9.9%) 33 (10.0%) Europe/West Asia 176 (53.3%) 179 (53.8%) 178 (53.9%) Asia and Pacific 29 (8.8%) 28 (8.4%) 27 (8.2%) *Lesion type determined by independent reading center
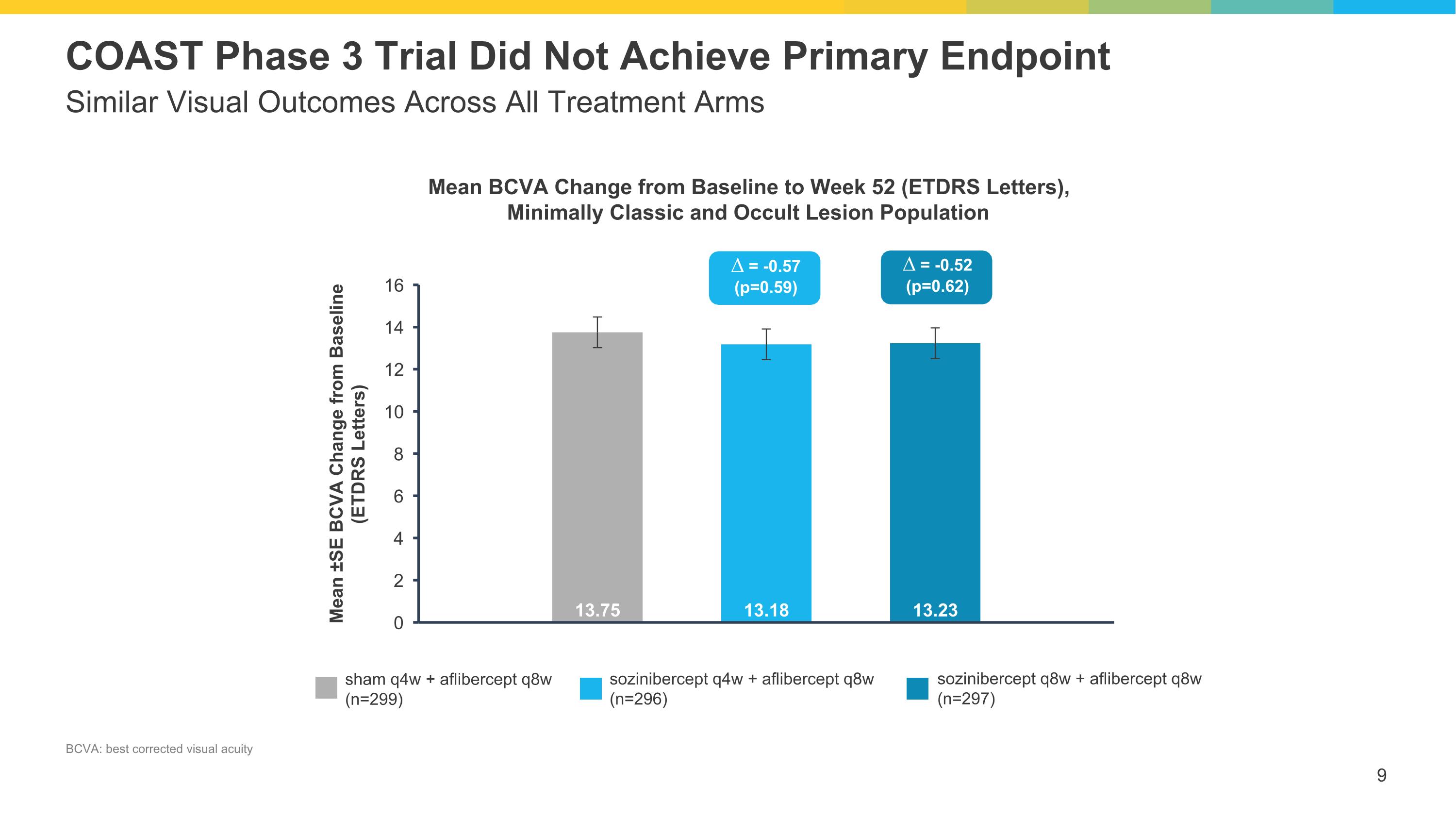
Similar Visual Outcomes Across All Treatment Arms COAST Phase 3 Trial Did Not Achieve Primary Endpoint sham q4w + aflibercept q8w (n=299) sozinibercept q4w + aflibercept q8w (n=296) sozinibercept q8w + aflibercept q8w (n=297) ∆ = -0.57 (p=0.59) ∆ = -0.52 (p=0.62) Mean BCVA Change from Baseline to Week 52 (ETDRS Letters), Minimally Classic and Occult Lesion Population BCVA: best corrected visual acuity
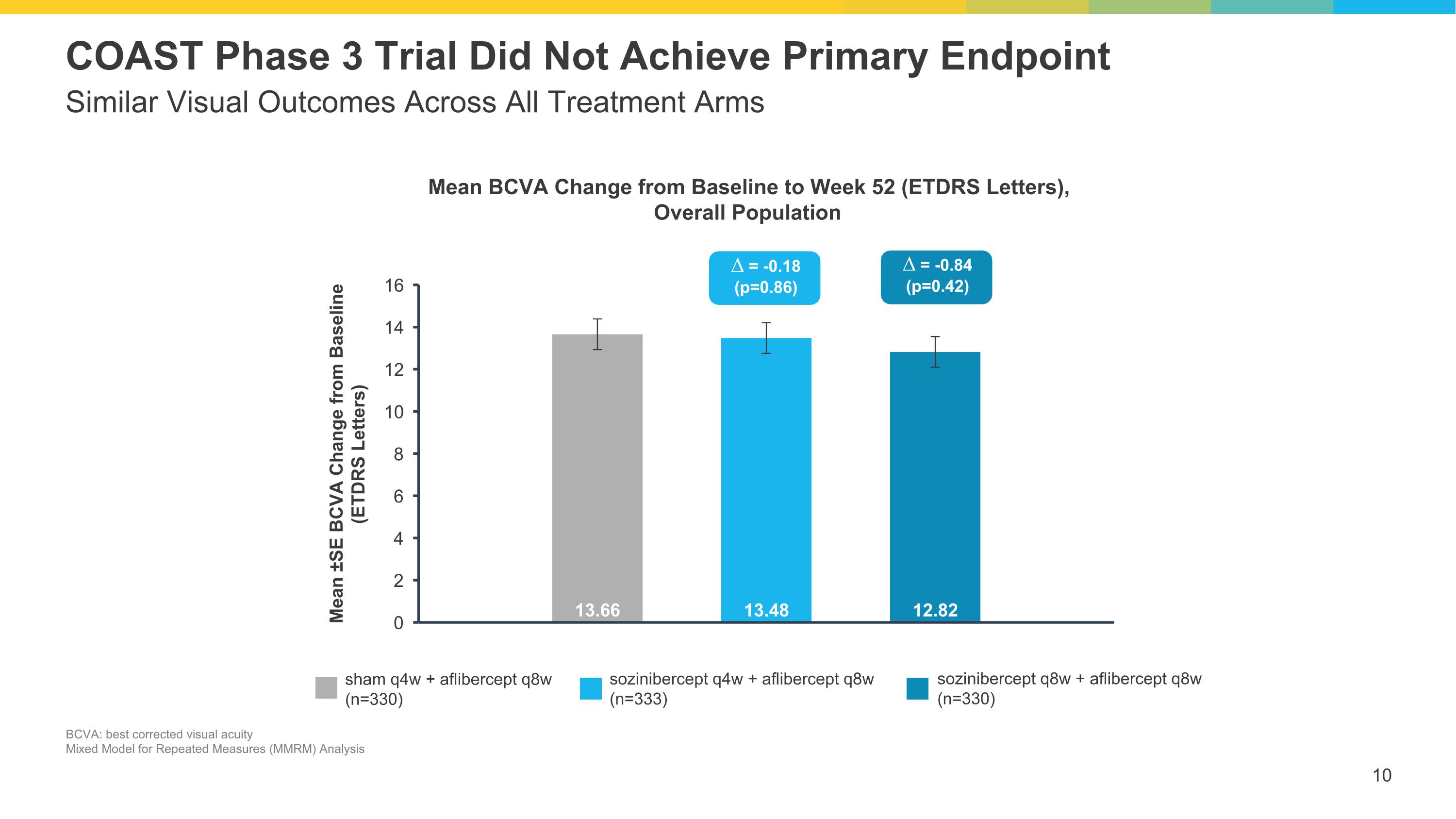
Similar Visual Outcomes Across All Treatment Arms COAST Phase 3 Trial Did Not Achieve Primary Endpoint sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) ∆ = -0.18 (p=0.86) ∆ = -0.84 (p=0.42) Mean BCVA Change from Baseline to Week 52 (ETDRS Letters), Overall Population BCVA: best corrected visual acuity Mixed Model for Repeated Measures (MMRM) Analysis
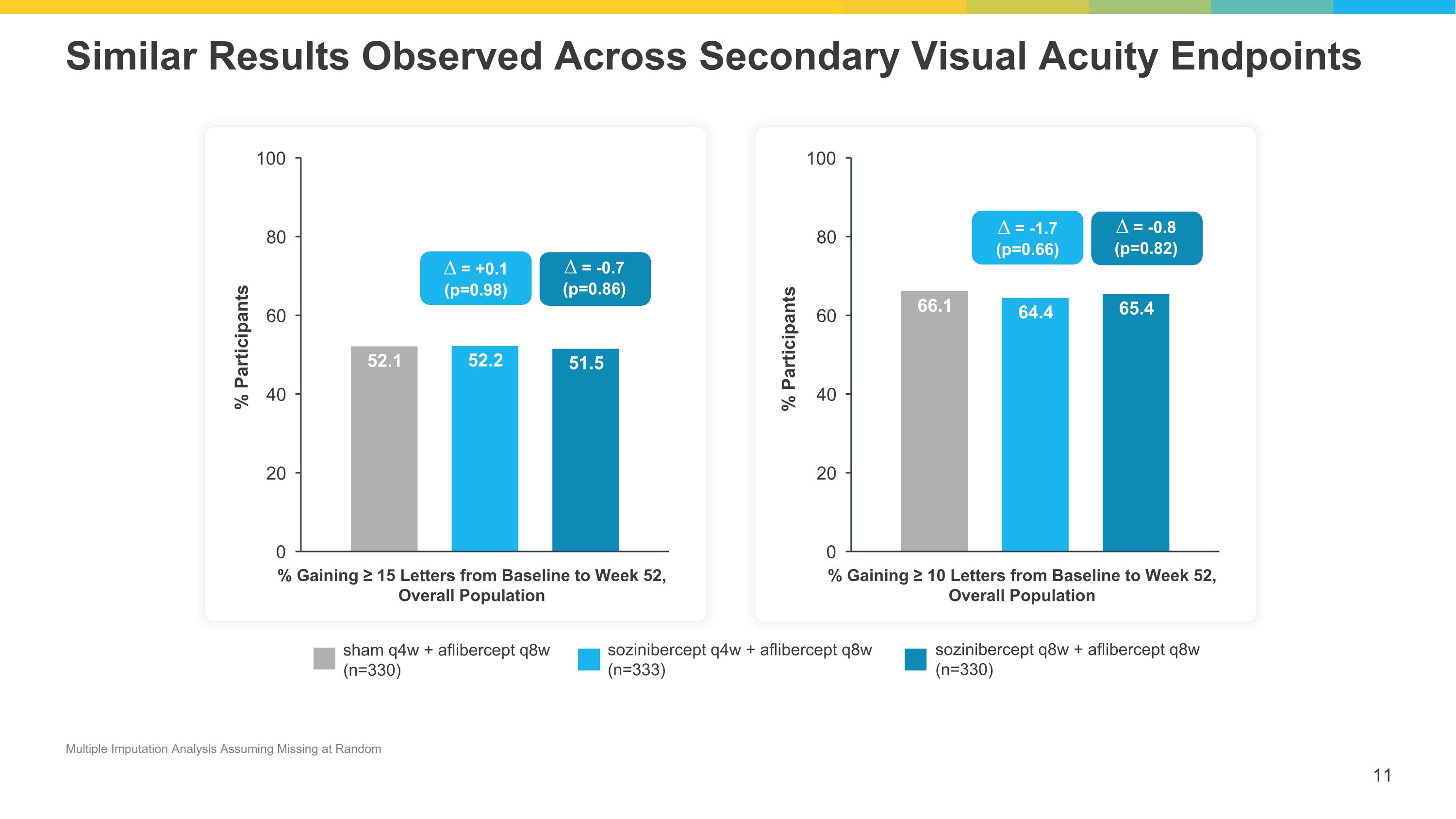
Similar Results Observed Across Secondary Visual Acuity Endpoints % Gaining ≥ 15 Letters from Baseline to Week 52, Overall Population % Gaining ≥ 10 Letters from Baseline to Week 52, Overall Population sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) ∆ = +0.1 (p=0.98) ∆ = -0.7 (p=0.86) ∆ = -1.7 (p=0.66) ∆ = -0.8 (p=0.82) Multiple Imputation Analysis Assuming Missing at Random
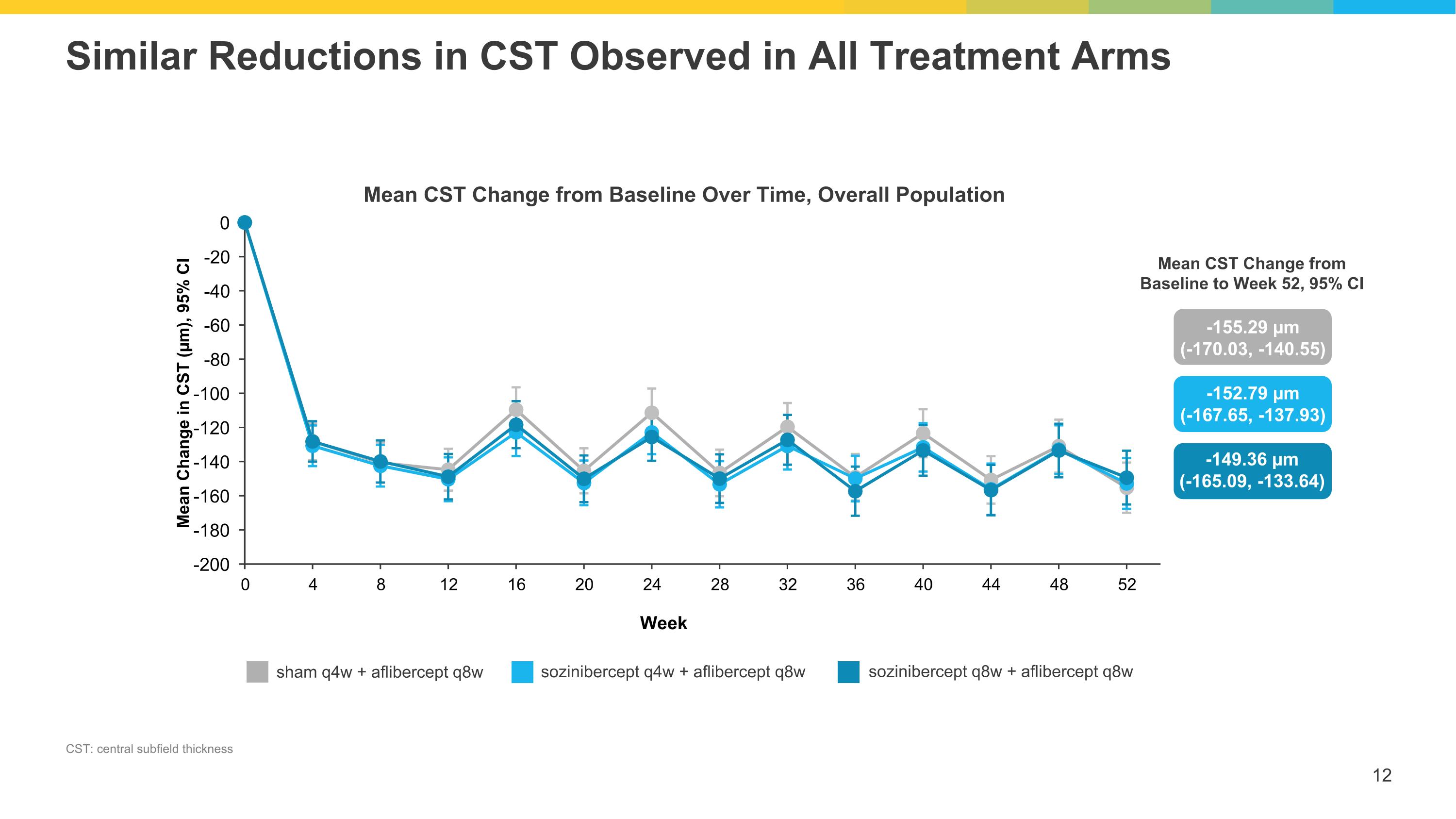
Similar Reductions in CST Observed in All Treatment Arms CST: central subfield thickness Mean CST Change from Baseline Over Time, Overall Population -155.29 µm (-170.03, -140.55) sham q4w + aflibercept q8w sozinibercept q4w + aflibercept q8w sozinibercept q8w + aflibercept q8w -152.79 µm (-167.65, -137.93) -149.36 µm (-165.09, -133.64) Mean CST Change from Baseline to Week 52, 95% CI
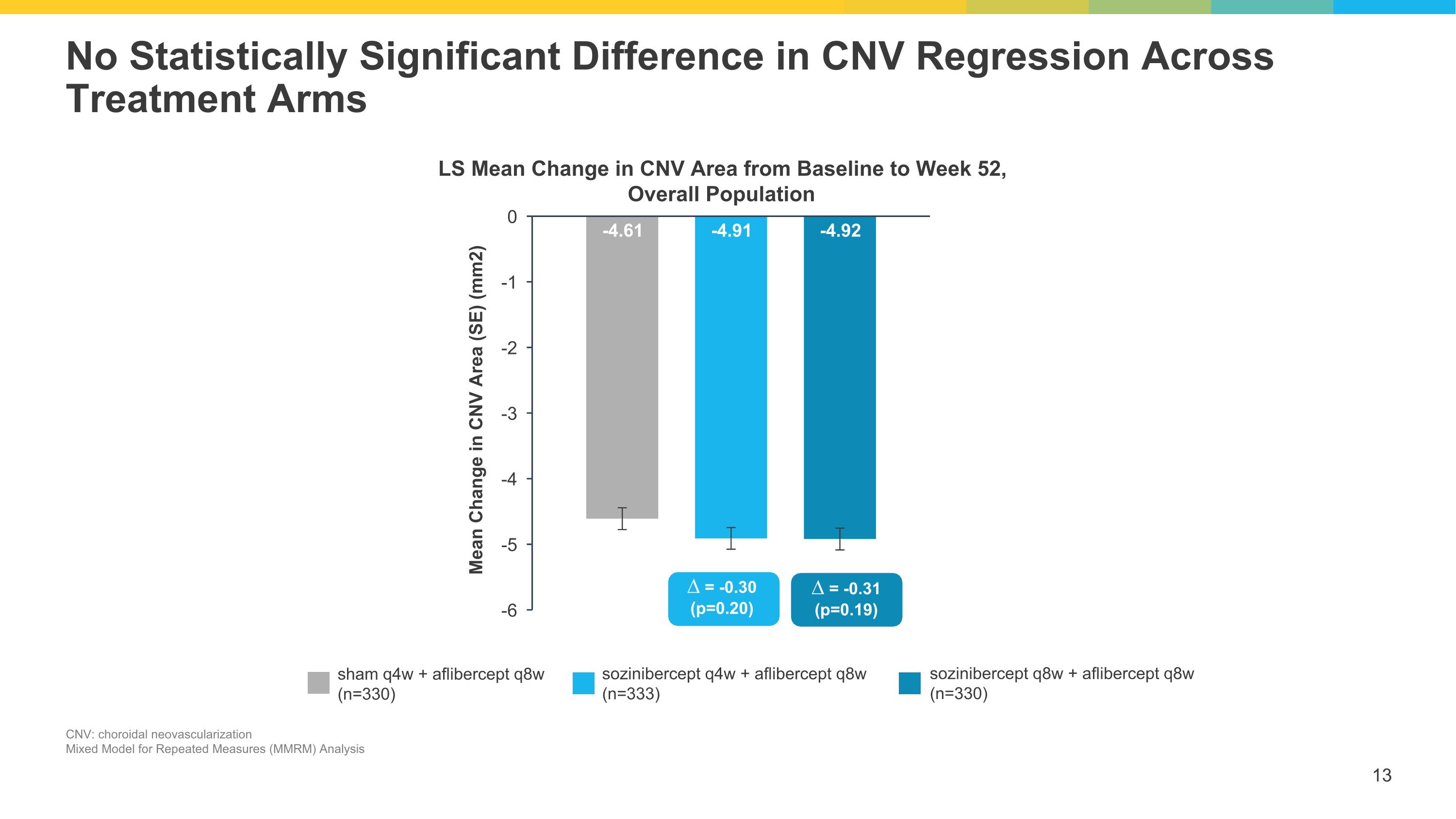
No Statistically Significant Difference in CNV Regression Across Treatment Arms CNV: choroidal neovascularization Mixed Model for Repeated Measures (MMRM) Analysis LS Mean Change in CNV Area from Baseline to Week 52, Overall Population sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) ∆ = -0.30 (p=0.20) ∆ = -0.31 (p=0.19)
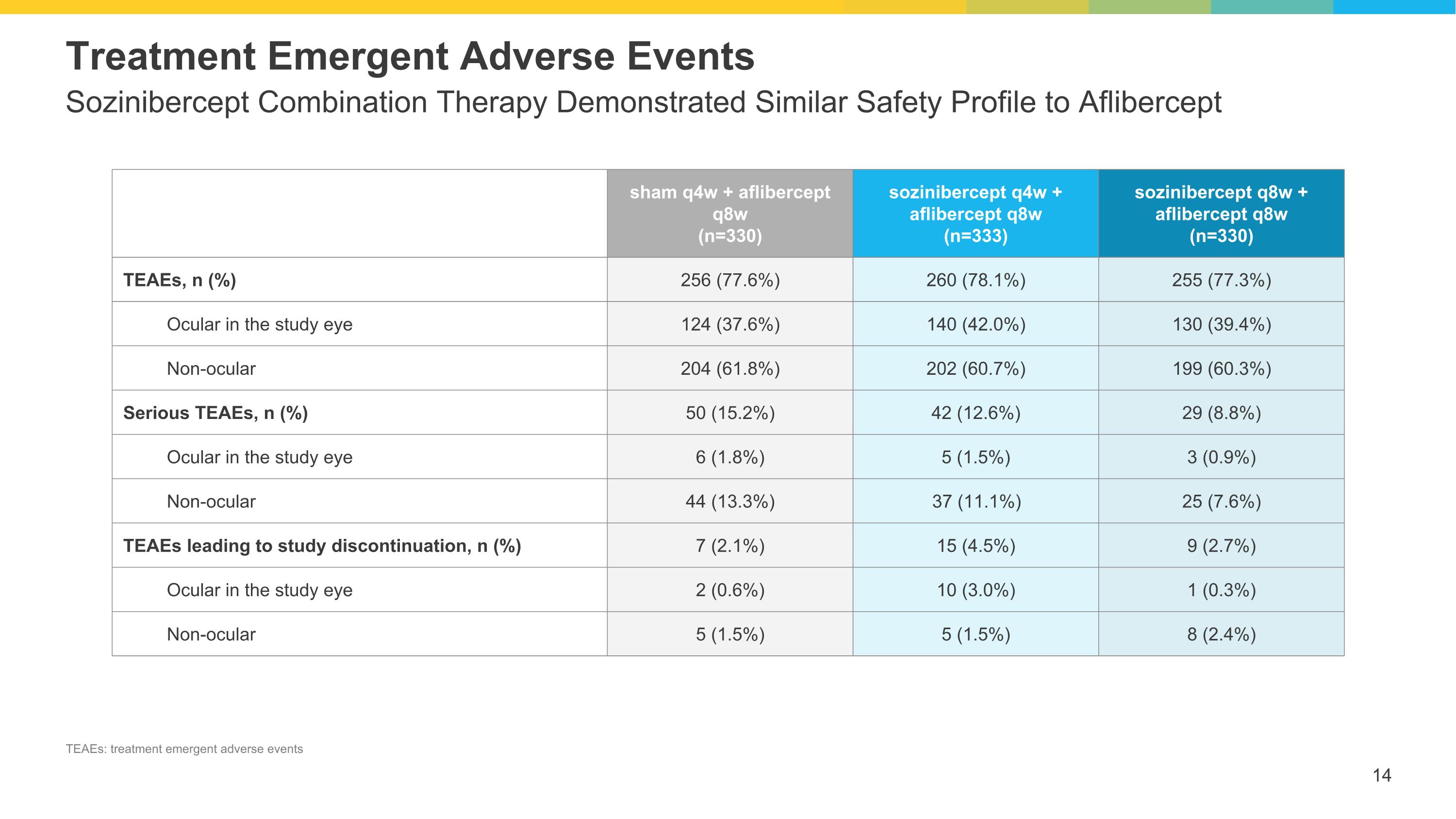
Sozinibercept Combination Therapy Demonstrated Similar Safety Profile to Aflibercept Treatment Emergent Adverse Events sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) TEAEs, n (%) 256 (77.6%) 260 (78.1%) 255 (77.3%) Ocular in the study eye 124 (37.6%) 140 (42.0%) 130 (39.4%) Non-ocular 204 (61.8%) 202 (60.7%) 199 (60.3%) Serious TEAEs, n (%) 50 (15.2%) 42 (12.6%) 29 (8.8%) Ocular in the study eye 6 (1.8%) 5 (1.5%) 3 (0.9%) Non-ocular 44 (13.3%) 37 (11.1%) 25 (7.6%) TEAEs leading to study discontinuation, n (%) 7 (2.1%) 15 (4.5%) 9 (2.7%) Ocular in the study eye 2 (0.6%) 10 (3.0%) 1 (0.3%) Non-ocular 5 (1.5%) 5 (1.5%) 8 (2.4%) TEAEs: treatment emergent adverse events
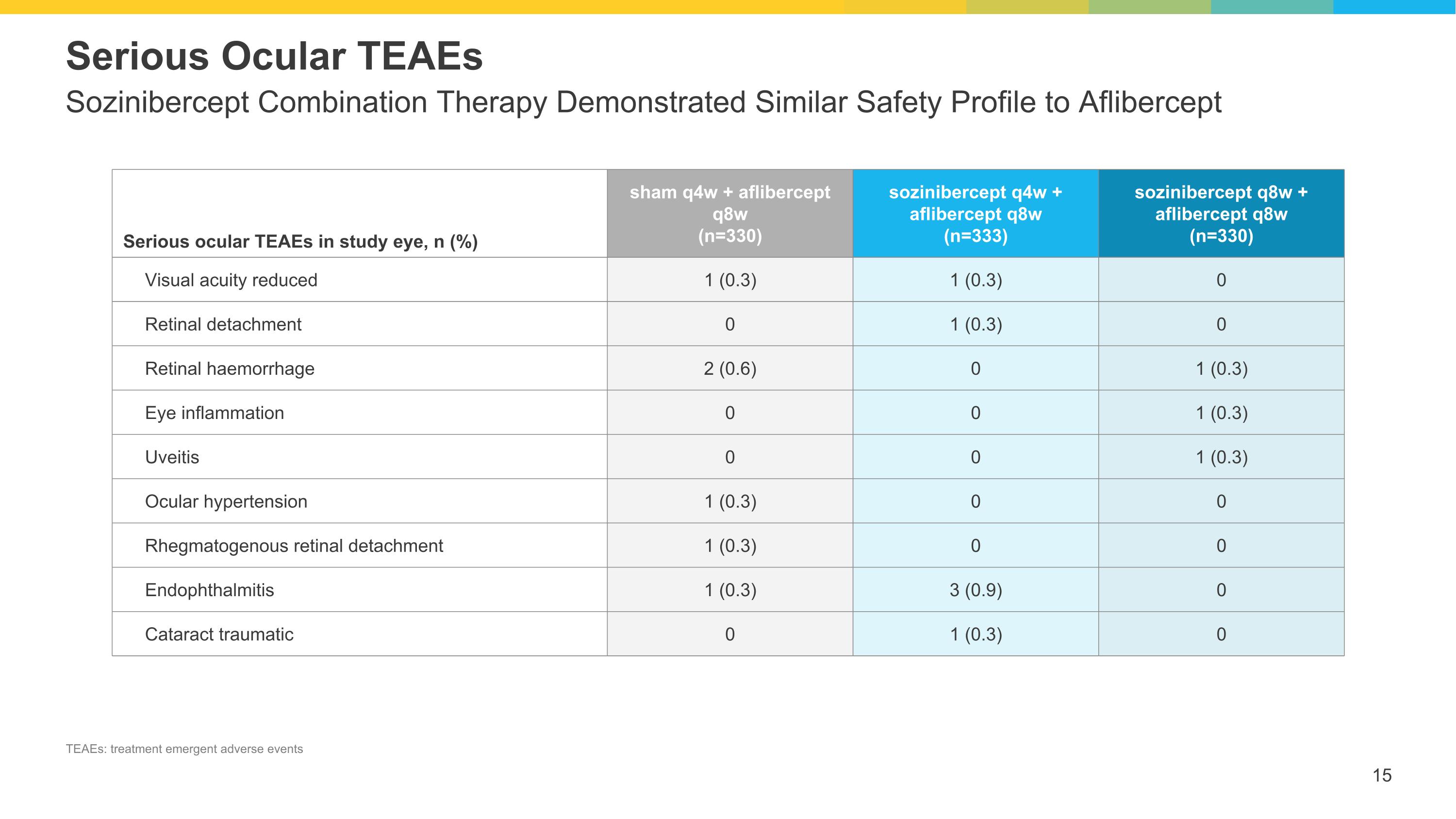
Sozinibercept Combination Therapy Demonstrated Similar Safety Profile to Aflibercept Serious Ocular TEAEs Serious ocular TEAEs in study eye, n (%) sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) Visual acuity reduced 1 (0.3) 1 (0.3) 0 Retinal detachment 0 1 (0.3) 0 Retinal haemorrhage 2 (0.6) 0 1 (0.3) Eye inflammation 0 0 1 (0.3) Uveitis 0 0 1 (0.3) Ocular hypertension 1 (0.3) 0 0 Rhegmatogenous retinal detachment 1 (0.3) 0 0 Endophthalmitis 1 (0.3) 3 (0.9) 0 Cataract traumatic 0 1 (0.3) 0 TEAEs: treatment emergent adverse events
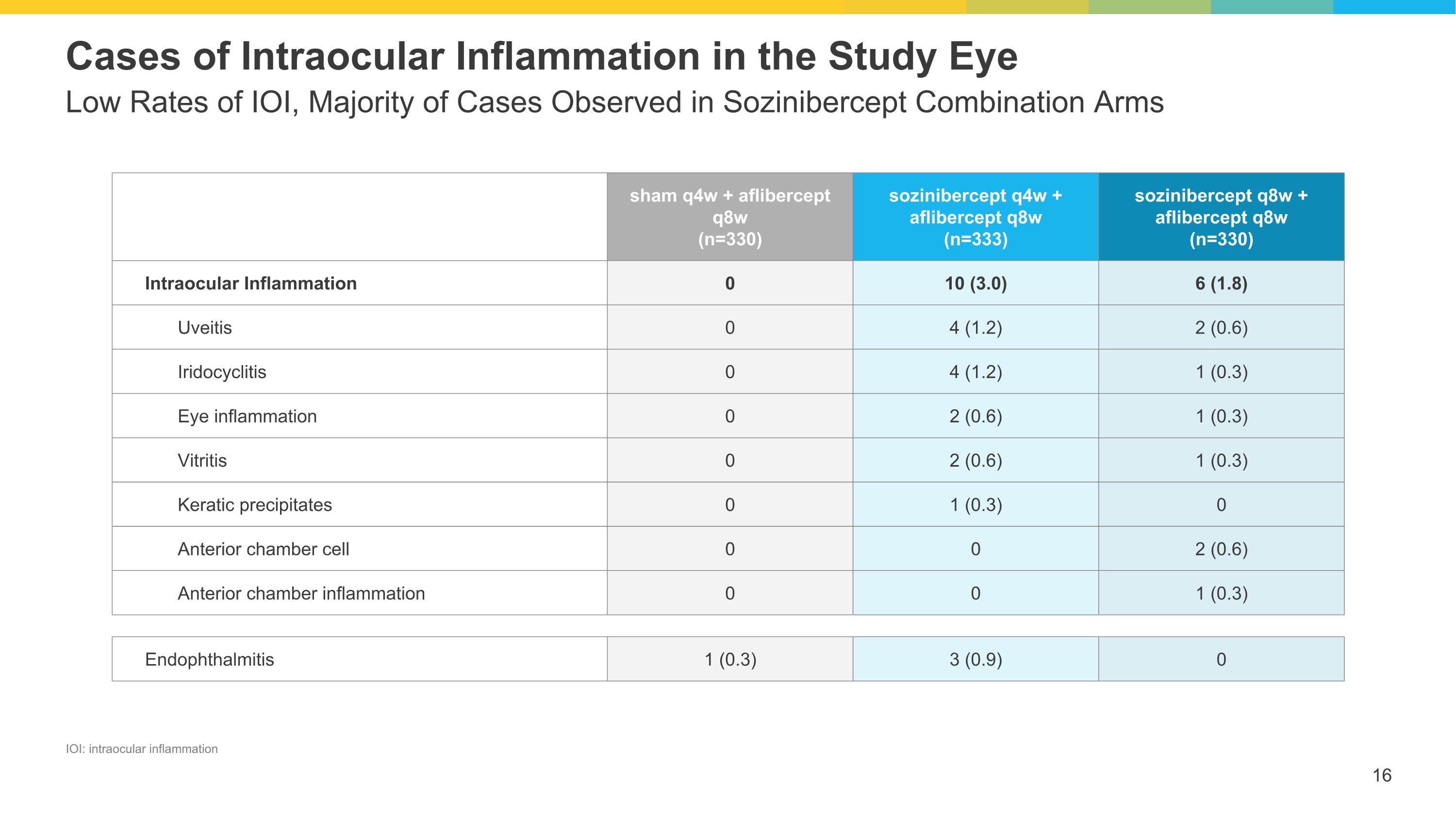
Low Rates of IOI, Majority of Cases Observed in Sozinibercept Combination Arms Cases of Intraocular Inflammation in the Study Eye sham q4w + aflibercept q8w (n=330) sozinibercept q4w + aflibercept q8w (n=333) sozinibercept q8w + aflibercept q8w (n=330) Intraocular Inflammation 0 10 (3.0) 6 (1.8) Uveitis 0 4 (1.2) 2 (0.6) Iridocyclitis 0 4 (1.2) 1 (0.3) Eye inflammation 0 2 (0.6) 1 (0.3) Vitritis 0 2 (0.6) 1 (0.3) Keratic precipitates 0 1 (0.3) 0 Anterior chamber cell 0 0 2 (0.6) Anterior chamber inflammation 0 0 1 (0.3) Endophthalmitis 1 (0.3) 3 (0.9) 0 IOI: intraocular inflammation
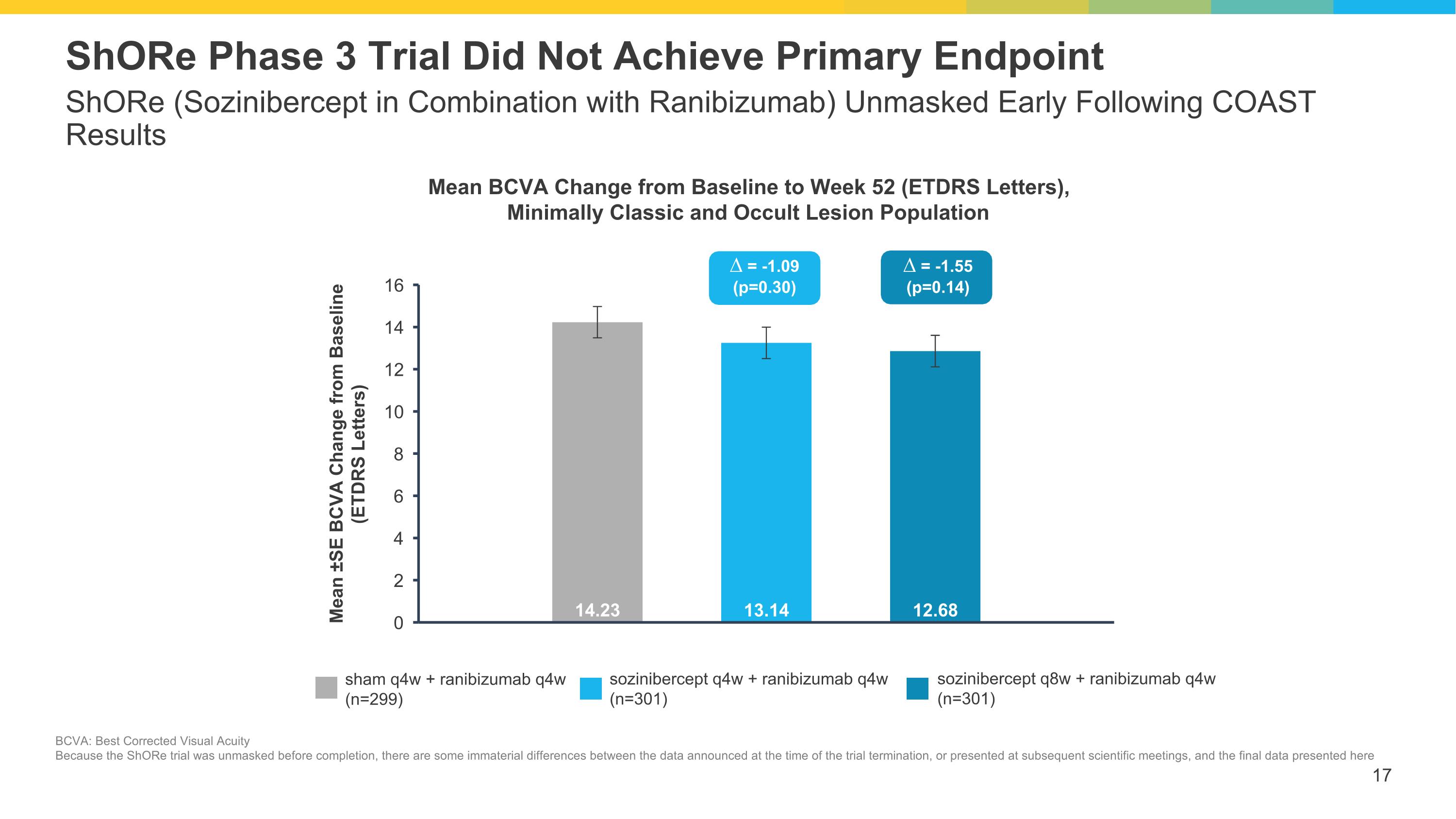
ShORe (Sozinibercept in Combination with Ranibizumab) Unmasked Early Following COAST Results ShORe Phase 3 Trial Did Not Achieve Primary Endpoint sham q4w + ranibizumab q4w (n=299) sozinibercept q4w + ranibizumab q4w (n=301) sozinibercept q8w + ranibizumab q4w (n=301) ∆ = -1.09 (p=0.30) ∆ = -1.55 (p=0.14) Mean BCVA Change from Baseline to Week 52 (ETDRS Letters), Minimally Classic and Occult Lesion Population BCVA: Best Corrected Visual Acuity Because the ShORe trial was unmasked before completion, there are some immaterial differences between the data announced at the time of the trial termination, or presented at subsequent scientific meetings, and the final data presented here
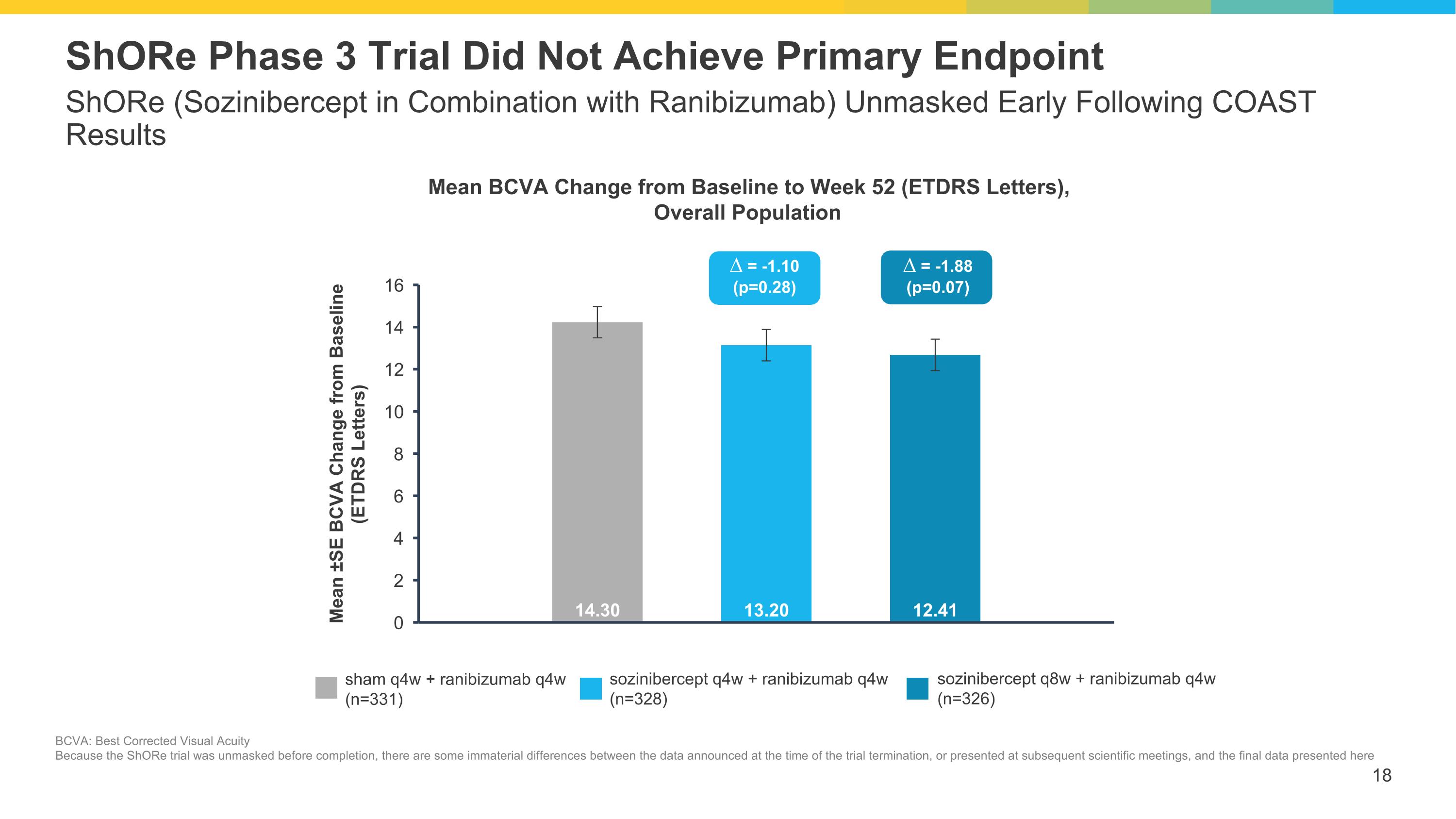
ShORe (Sozinibercept in Combination with Ranibizumab) Unmasked Early Following COAST Results ShORe Phase 3 Trial Did Not Achieve Primary Endpoint sham q4w + ranibizumab q4w (n=331) sozinibercept q4w + ranibizumab q4w (n=328) sozinibercept q8w + ranibizumab q4w (n=326) ∆ = -1.10 (p=0.28) ∆ = -1.88 (p=0.07) Mean BCVA Change from Baseline to Week 52 (ETDRS Letters), Overall Population BCVA: Best Corrected Visual Acuity Because the ShORe trial was unmasked before completion, there are some immaterial differences between the data announced at the time of the trial termination, or presented at subsequent scientific meetings, and the final data presented here
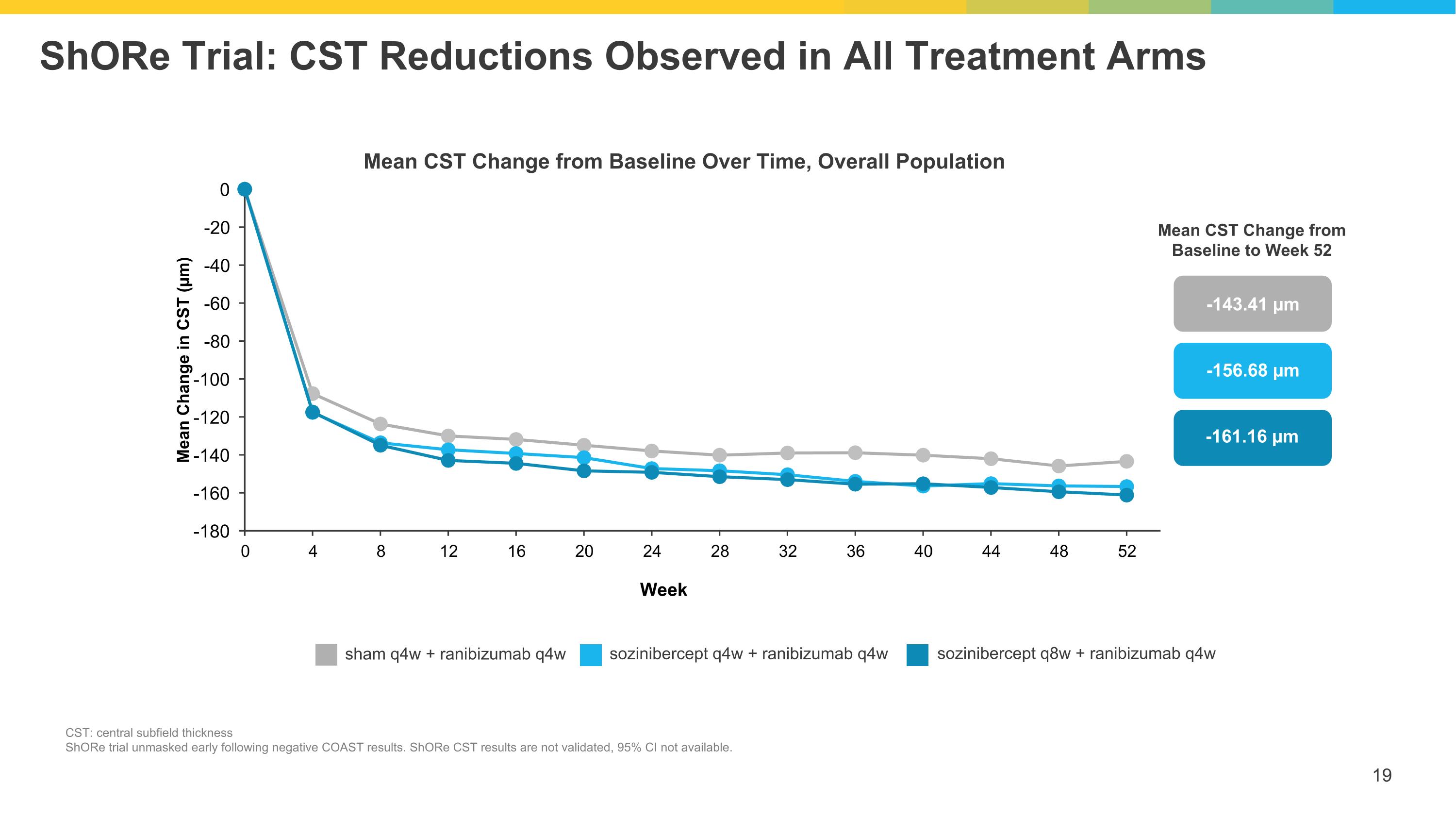
ShORe Trial: CST Reductions Observed in All Treatment Arms CST: central subfield thickness ShORe trial unmasked early following negative COAST results. ShORe CST results are not validated, 95% CI not available. Mean CST Change from Baseline Over Time, Overall Population -143.41 µm -156.68 µm -161.16 µm Mean CST Change from Baseline to Week 52 sham q4w + ranibizumab q4w sozinibercept q4w + ranibizumab q4w sozinibercept q8w + ranibizumab q4w

The COAST and ShORe Phase 3 trials did not meet the primary endpoint of superiority in vision improvement from baseline to week 52 No statistically significant differences in key anatomical outcomes were observed between treatment arms Sozinibercept combination therapy was well tolerated In this large global Phase 3 program, VEGF-C and VEGF-D inhibition did not provide additional functional benefit beyond standard of care anti-VEGF-A therapy in wet AMD patients COAST and ShORe Trials Key Takeaways

Update on Development Funding Agreement (DFA) Negotiations��Tom Reilly

In August 2022, Opthea entered into a Development Funding Agreement (DFA) with Ocelot SPV LP which provided USD$120M of funding to support our development of sozinibercept for the treatment of wet AMD In December 2023, Opthea entered into an Amended and Restated DFA with Ocelot as collateral agent, pursuant to which a new co-investor provided an additional USD$50 million in funding, bringing the total funding to USD$170 million If sozinibercept was approved, repayment to the DFA investors was 4x investment (USD$680M) Under the DFA, the DFA investors held security over the assets of Opthea in the form of an "all assets" lien There are termination clauses in the DFA which involve repayments ranging from USD$0M up to USD$680M Original terms of the DFA

Following the negative topline data of the COAST trial, Opthea’s management and Board of Directors began discussions with the DFA investors related to the next steps of the Sozinibercept Wet AMD program In consultation with the DFA investors, Opthea determined the most appropriate course of action for wet AMD patients, Opthea shareholders, and other stakeholders was to accelerate the timing of the ShORE trial topline data Following the negative topline data of the COAST and ShORE trial, Opthea in consultation with the DFA investors determined to terminate the Sozinibercept wet AMD program. Opthea determined that this was in the best interests of Opthea shareholders, including to preserve cash Since these negative Phase 3 results, Opthea has reduced the work force by over 80%, reduced the Board of Directors by 50%, renegotiated all contracts related to the clinical trials & had active discussions with the DFA investors to settle the DFA arrangements As announced on August 18th/August 19th (Australia), Opthea has agreed to settle with the DFA investors with a cash payment of USD20M & 9.99% equity stake in the Company (equivalent to 136.7M common shares). These settlement arrangements include termination of the DFA and all liens(1) As of August 19th, 2025 (after payment of USD20M to the DFA investors) the company has cash of approximately USD20M and after issue of the new shares to the DFA investors ~1,4B common shares outstanding Successful DFA Negotiations Allowed The Company to Remain Solvent Which Is a Better Outcome For Shareholders (1) For further detail regarding the material terms of the Settlement Agreement and Subscription Deed, see the Company's ASX Announcement dated August 19th, 2025

Current State of the Company��Dr Jeremy Levin

Opthea settled arrangements with the DFA investors removing financial uncertainty Opthea retains meaningful assets: Significant cash position No debt No lien on any asset (1) Dual-listing on ASX and NASDAQ. Trading in Opthea's listed securities remains suspended by ASX under ASX Listing Rule 17.3. Opthea is currently engaging with the ASX regarding these matters. Clinical, preclinical, scientific knowledge and assets and IP related to VEGF-C and VEGF-D Existing API and materials to allow potential new investigations Streamlining of operations has been achieved. Only 3 employees to remain as of September 15th Board of Directors was reduced by half to four directors. Sujal Shah to step down as of September 15th. As announced to the market on August 19th, given settlement of the DFA arrangements, Opthea is no longer relying on the ‘safe harbour’ provisions in section 588GA of the Corporations Act 2001 (Cth). Successful DFA Negotiations Resulted in Positive Outcome for Shareholders (1) For further detail regarding the material terms of the Settlement Agreement and Subscription Deed, see the Company's ASX Announcement dated August 19th 2025

The Path Forward��Dr Jeremy Levin

Board of Directors: • Dr. Jeremy Levin • Kathy Connell • Lawrence Gozlan • Sujal Shah, who will step down as of September 15th, 2025 Planned Executive Departures: • Fred Guerard, CEO – stepping down effective September 1st, 2025 • Tom Reilly, CFO – stepping down effective September 15th, 2025 • Karen Adams, Corporate Secretary – stepping down effective November 1, 2025 Management: We have implemented a streamlined, cost-efficient structure that aligns with the Company’s current scale and strategic priorities. This includes active engagement of our Board, whose deep expertise across areas including science, business development, finance, commercial operations, and investment is being fully leveraged to ensure effective governance, operational oversight Leadership and Governance

The Board will continue to focus on maximizing shareholder value and will assess the following: Full strategic review over the next six months Targeted internal development Strategic partnerships or potential BD/licensing, where appropriate Return of capital to shareholders, where appropriate Strategy To Maximize Shareholder Return

Strategic Transition: Clear Priorities, Steady Progress A comprehensive business and asset review is actively underway The Board Is focused on delivering long-term shareholder value Will provide additional support to the company during these transitional stages Expects to provide shareholders with a further update in CY Q4 Thank you for your continued trust and support Executing with Focus and Accountability

Q&A

Thank you































